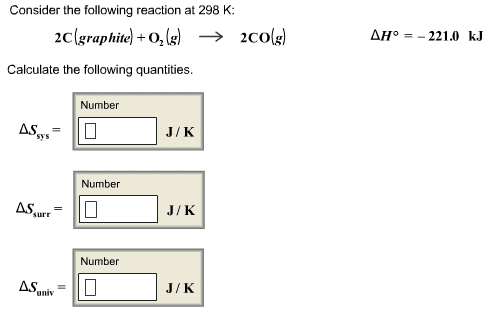
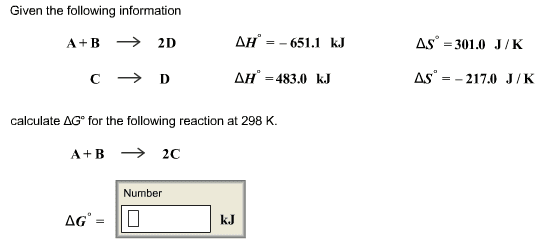
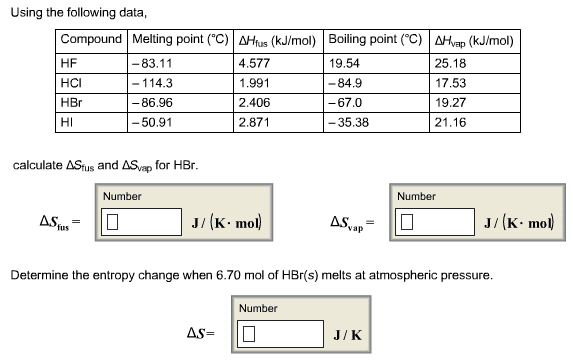
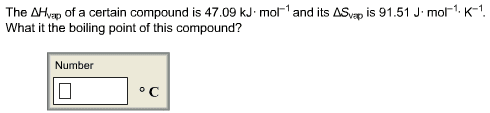please help and explain... I am beyond lost on this assignment
1. A certain process has âHâ¦>0 and âSâ¦<0. Which of the following is a correct conclusion about this process
Non spontaneous at all T.
Spontaneous at high T / Spontaneous at low T / Spontaneous at all T / None of the aboveÂÂÂÂÂÂÂÂÂÂÂÂÂÂÂÂÂÂÂ
2. the formation constant for the reaction Ag+(aq)+2NH3(aq) Ã-à Ag(NH3)2+(aq) is K=1.7x10^7 at 25 deg C. what is âG⦠(kJ/mol) at this temp
-23 / -18 / -3.5 / -1.5 / -41
3. Nitric oxide reacts with chlorine to form NOCL. What is the free energy (kJ) of the reaction at 277 dec C.
2NO(g) + Cl2(g) Ã 2NOCl(g)
+144 / -41.0 / -10.3 / -22.2 / +41.0
4. How much free energy is released or absorbed(kJ) when 95.0 g of ozone is produced.
3 O2(g)à 2 O3(g) âG⦠= +326 kJ
+645 / -323 / +95 / +323 / -645
5. Estimate the boiling point (deg C) of phosphoric acid? H3PO4(s)ÃÃ H3PO4(l)
42 / 181 / 315 / -92 / 305
6. Calculate the âS⦠(J/K) for the reaction, SiCl4(g)+2Mg(s)à 2MgCl2(s)+Si(s)
+198 / -198 / +255 / -255 / +472
7. Calculate the equilibrium constant, K, for the following reaction at 25 deg C: CH4(g)+2H2O(g)Ãà CO2(g) âG⦠=-113.6kJ
Which of the following results in a decrease in the entropy of the system
O2(g),300KÃ O2(g), 400K / H2O(s)Ã H3O(l) / N2(g)Ã N2(aq) / NH3(l)Ã NH3(g) / 2H2O2(g)Ã 2H2O(g)+O2(g)
8. Identify the incorrect statemtent?
When âG⦠>0, energy can be added to force a non-spontaneous to proceed.
When K>1, âG⦠<0.
Enthalpy is a good predictor of free energy.
Entropy of the universe increases in a spontaneous reaction
All of the above