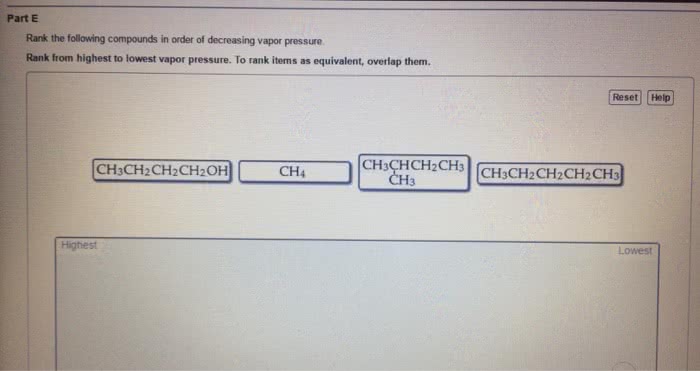
CHEM 130 Lecture Notes - Lecture 33: Boiling Point, Dipole, Intermolecular Force
Document Summary
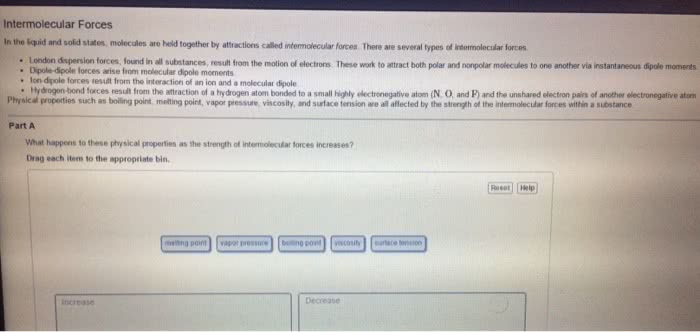
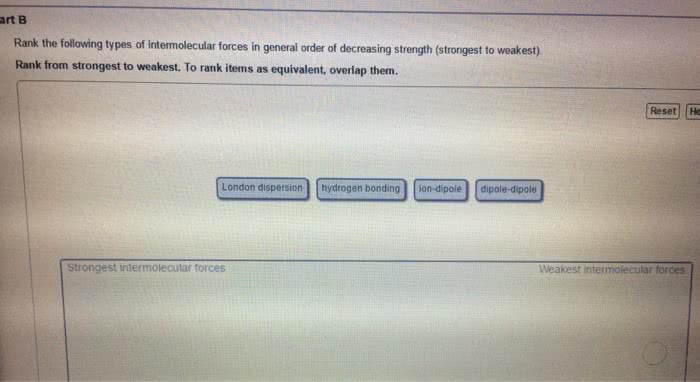
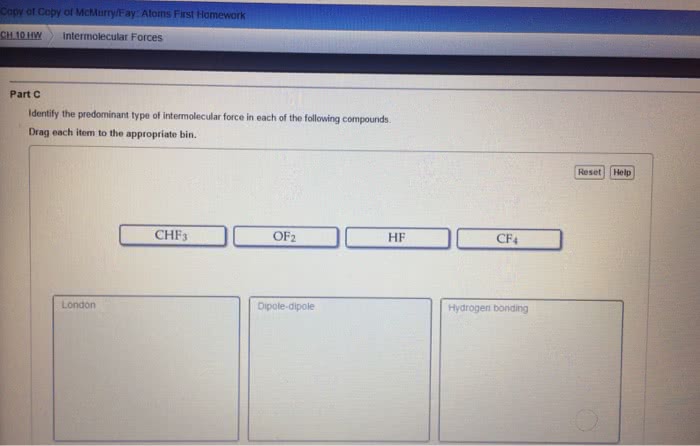
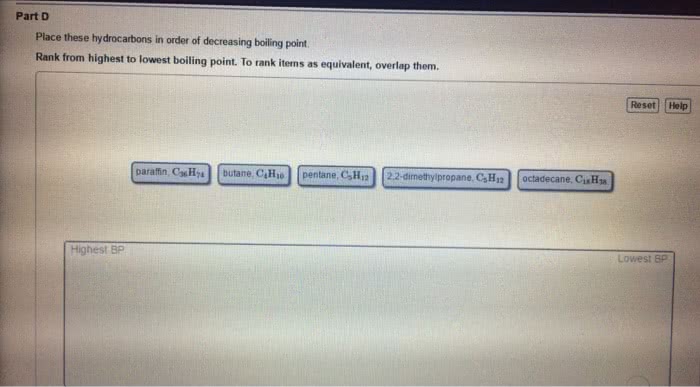
Chem 130: general chemistry: macroscopic investigations and reaction principles - Three types of attractive forces known to exist between neutral molecules. An attractive intermolecular force resulting from the tendency of polar molecules to align themselves such that the positive end of one molecule is near the negative end of another. The weak attractive forces between molecules resulting from the small, instantaneous dipoles that occur because of the varying positions of the electrons during their motion about nuclei. Because molecules with larger molecules mass usually have more electrons and london forces increase in strength with the number of electrons. Larger molecular mass often means larger atoms, which are more polarizable. A weak to moderate attractive force that exists between a hydrogen atom covalently bonded to a very electronegative atom, x, and a lone pair of electrons on another small, electronegative atom, y. Occurs in substances containing hydrogen atoms bonded to certain very.