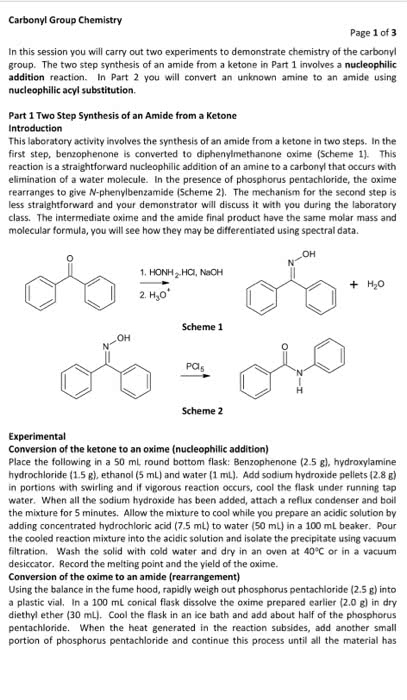
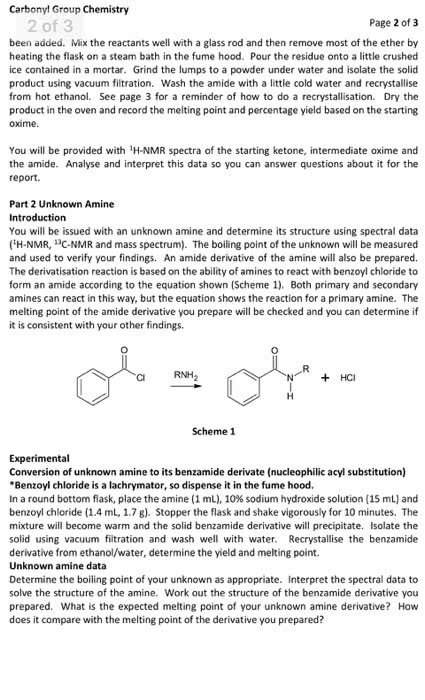
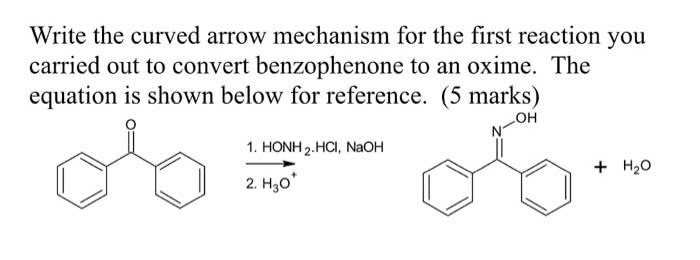
Please answer the prelab questions only that are 1,2 and 3. Thankyou
Using models may be helpful in answering these questions. If the brominating of room -still bone proceeded by seen addition what isomer(s) of stolen bromide would be formed? If the brominating of trans-stolen proceeded by anti-addition what isomer(s) of stolen bromide would be formed? Which isomer(s) of stolen bromide would be formed if the reaction followed Mechanism 4? Bromine is very toxic and corrosive. It can cause severe chemical burns on skin contact. Bromine vapor is very harmful. Make all transfers of bromine in the hood and if any is spilled, contact the instructor at once. Carry out the brominating part of this experiment in a hood. There is room to allow 2-3 people to work at each hood. Add about 2 g of trans-stolen to a thoroughly dried 125-ml. Erlenmeyer flask containing a dry magnetic stir bar. Add 20 mL of methylene chloride to the flask and swirl until the solid has dissolved. Clamp your flask to a ring stand over a magnetic stirrer. Adjust the stirring rate so that it is vigorous but without splashing. Obtain about 6 mL of a 2 M bromine solution in methylene chloride (approximately 10% bromine by volume) in a test tube. (Your instructor will provide the prepared solution.) A graduated cylinder will be placed in the hood to let you measure the bromine solution, this will avoid taking bromine-contaminated glassware out of the hood or to your bench. Once you have obtained your bromine solution, place the test tube in a small Kroc mayor flask for support and take it to your set-up in the hood. Carefully add about 1/3 of your bromine solution to your flask and note what color changes occur. Once the characteristic red-brown color of bromine has faded, add another third of your bromine solution to the flask. Again, note what changes occur. After the color has faded, add the remaining bromine solution. This lime, the color of bromine should persist for 3-5 minutes and not fade. If the color persists, the reaction is completed, and the color is due to excess bromine in the solution. If the color still fades, the reaction is not quite complete. If this is the case, add 10 drops of bromine solution and continue stirring until the color persists. A considerable amount of precipitate (the crude product) should be visible by this time. It may be difficult to determine if you have an excess of bromine. Allowing the material to settle for a minute or two should help you determine whether there is excess bromine in the solvent. Once you have a solution containing a small amount of excess bromine, add 1-2 drops of cyclohexane. This should cause the bromine color to disappear quickly. If the color still persists, add cyclohexane one drop at a time until the color fades, taking care not to add too much. Once the bromine color is gone, suspend the flask in an ice bath for 10-15 minutes, then recover the product by vacuum filtration on the Buchner funnel. Rinse the flask and product with two 3-ml. portions of ice-cold methylene chloride. Allow air to pass through your solid for at least 10 minutes to give your product lime to dry. Remove the solid from the Buchner funnel, and allow it to air dry for 10 minutes. Obtain the mass of your isolated product and determine its melting point. The melting point of racemic stilbene dibromide is 112-113 degree C. The melting point of meso-stilbene dibromide is 241-243 degree C. Calculate the yield of your product. You may be asked to obtain an IR spectrum for your product. Submit your product in an appropriately labeled vial.