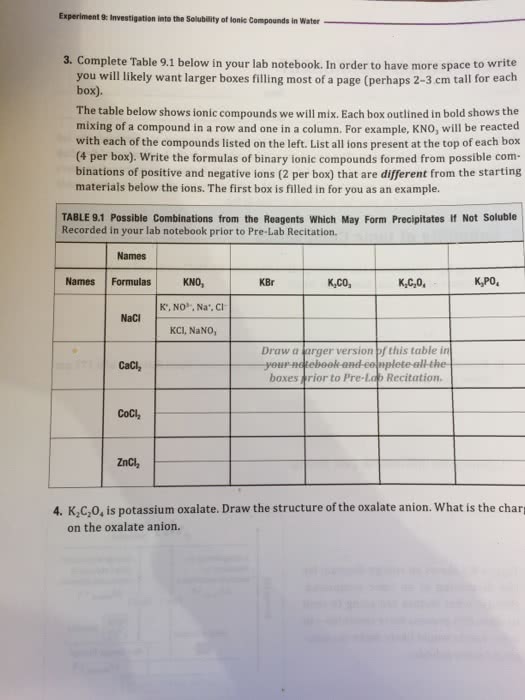
t 9 Investigation into the Solubility of lonic Compounds in Water 3. Complete Table 9.1 below in your lab notebook. In order to have more space to write you will likely want larger boxes filling most of a page (perhaps 2-3 cm tall for each box). mixing of a compound in a row and one in a column. For example, KNO, will be reacted with each of the compounds listed on the left. List all ions present at the top of each box (4 per box). Write the formulas of binary ionic compounds formed from possible com- binations of positive and negative ions (2 per box) that are different from the starting materials below the ions. The first box is filled in for you as an example. TABLE 9.1 Possible Combinations from the Reagents Which May Form Precipitates If Not Soluble Recorded in your lab notebook prior to Pre-Lab Recitation. Names Names Formulas KNO, KBr K,CO, K,PO K', NO3, Na, ci NaCI KCI, NaNO, Draw a arger version of this table in your notebook-and ce boxes rior to Pre-Lab Recitation. CaCl, ZnC 4·K,Go, is potassium oxalate. Draw the structure of the oxalate anion, what is the char on the oxalate anion.