CHEM 2332H Lecture : Lecture 492014.pdf
Document Summary
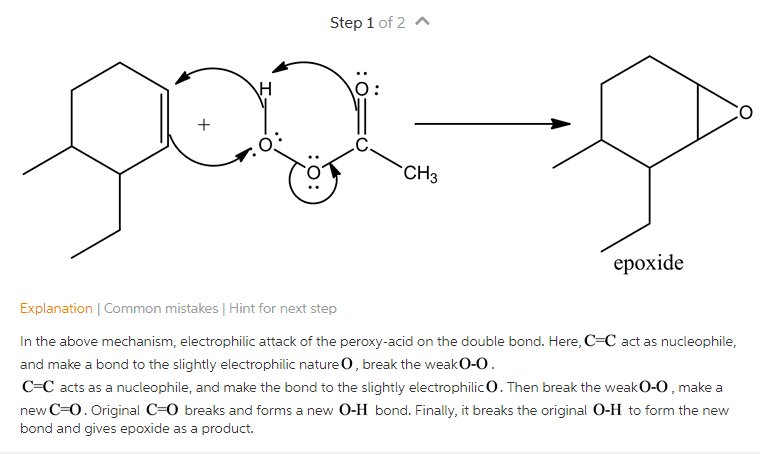
B/c o is positive, becomes electrophilic for even a weak nucleophile like cl- to attack. When chloride attacks as nucleophile, is the better leaving group but gas formation step after that isn"t eqbm, so that pulls rxn forward (even though just for cl attack, eqbm might lie towards reactants) Good for acyl substitution rxns (chloride = leaving group) Can replace cl by h (reduction) or by c (organoreduction/friedel-crafts acylation) also works w/ secondary amines. Synthesis practice problems ex: 7 c or less (no heteroatoms) can"t use esterification here right away (fischer esterification) Ochem page 2 can"t use esterification here right away conc. Can use the one we just learned today as well but now need 2 rxns. Functional group interconversion acid --> ester can also use fischer esterification want to put them in w/o adding other groups. Na2cr2o7 these are our 2 choices ring is better b/c quicker want antimarkovnikov hydration herehydroboration: bh3. thf, h2o2.