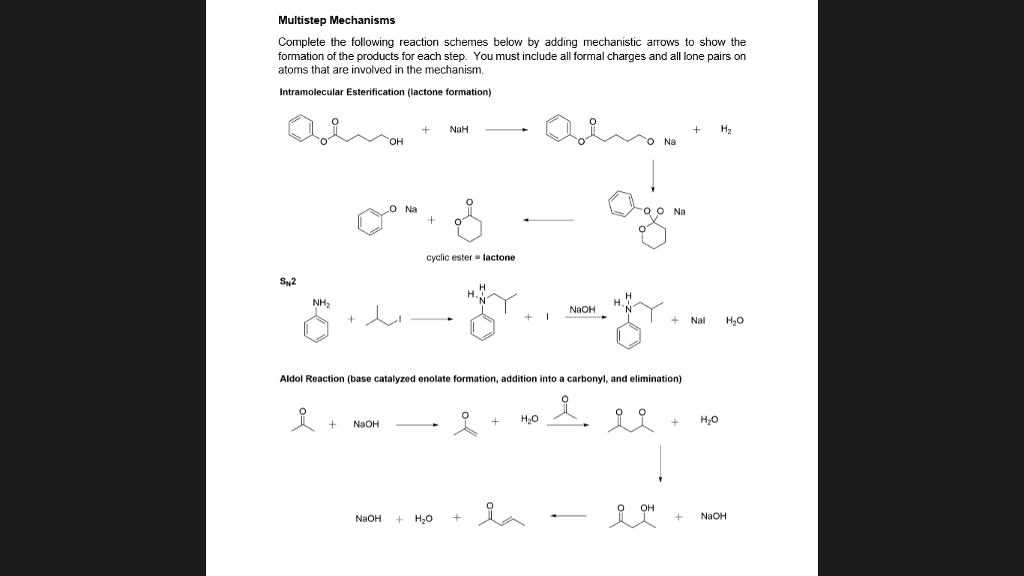
CHEM 2332H Lecture Notes - Aryne, Electrophile
17 views6 pages
15 Jul 2014
School
Department
Course
Professor
Get access
Grade+20% off
$8 USD/m$10 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
40 Verified Answers
Class+
$8 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
30 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232
Related Documents
Related Questions
1.Which of the following statements are true about sulfuricacid's role in the esterification reaction. Note thatthere may be more than one correct answer.
| sulfuric acid is added to lower the boiling point of thereaction mixture. | ||
| sulfuric acid protonates the carboxylic acid in the first stepof the reaction mechanism | ||
| sulfuric acid is used to ensure the alcohol will dissolve inether | ||
| sulfuric acid protonates the ester in the first step of thereaction mechanism | ||
| sulfuric acid protonates the alcohol in the first step of thereaction mechanism | ||
| sulfuric acid reduces the concentration of OH- andslows the reverse reaction. | ||
| sulfuric acid is used to ensure the carboxylic acid willdissolve in ether | ||
| sulfuric acid is added to react with excess sodium bicarbonatein the first step of the reaction | ||
| sulfuric acid acts as a catalyst for the reaction |
2.Which of the following below explain why the alcohol is usedin excess in this reaction. Note that there may be morethan one answer.
| |||||||||||||||||||||||
3.Why is a sodium bicarbonate solution used to extract the ether(organic) layer. Note that there may be more than onecorrect answer.
| sodium bicarbonate reacts with the ester to make it soluble inwater. | ||
| sodium bicarbonate should not be added, sulfuric acid should beadded during the extraction. | ||
| sodium bicarbonate reacts with any leftover sulfuric acid toform a salt that is INsoluble in the organic layer. | ||
| sodium bicarbonate reacts with the alcohol to make it solublein water. | ||
| sodium bicarbonate is added because it is less expensive thanadding hydrochloric acid. | ||
| sodium bicarbonate reacts with any leftover carboxylic acid toform a salt that is INsoluble in the organic layer. | ||
| sodium bicarbonate reacts with any leftover carboxylic acid toform a salt that is soluble in the organic layer. | ||
| sodium bicarbonate is used to react with ether and make theyield of the ester product greater. | ||
| sodium bicarbonate reacts with any leftover sulfuric acid toform a salt that is soluble in the organic layer. |