CHEM 102 Lecture Notes - Lecture 11: Mole Fraction, Partial Pressure, Molar Concentration
Document Summary
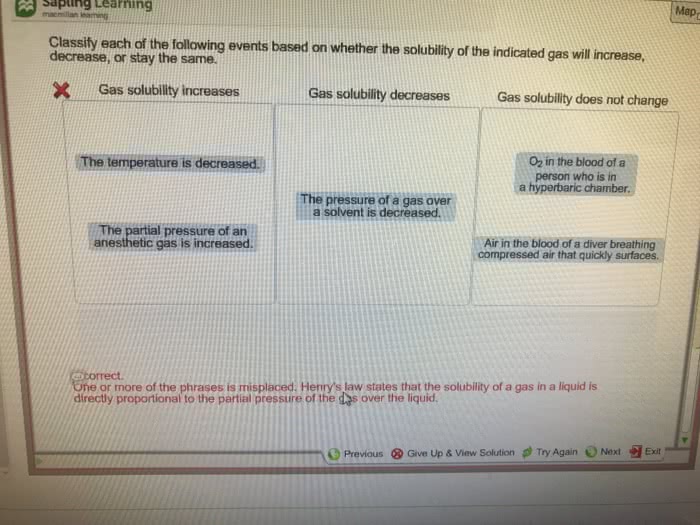
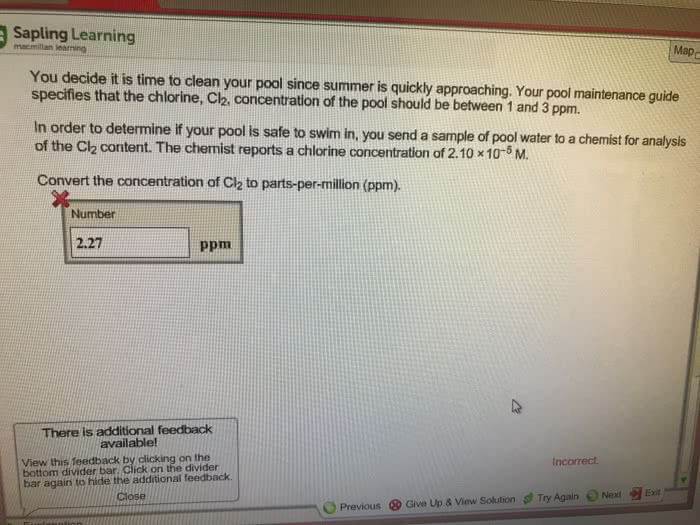
Chem 102: general descriptive chemistry ii - lecture 11: gas concentration, The henry"s law constant in this situation is 3. 4 x 10-2 mol/l x atm: sg = (3. 4 x 10-2 mol/l atm) x (3. 0 x 10-4 atm, answer: 1. 02 x 10-5 mol/l. Temperature and gases in solution: for gases in liquid, solubility decreases with increasing temperature, carbonated soft drinks more bubbly in the refrigerator. What is the molality of glucose: answer: 0. 964 m. If you know the density of solution, you can calculate molality from the molarity and vice versa. Colligative properties: colligative property a property that is dependent only on the number of solute particles present, not the identity of solute particles. Four colligative properties: vapor pressure lowering, boiling point elevation, freezing point depression, osmotic pressure. Vapor pressure lowering: because of solute-solvent im attraction, higher concentrations of nonvolatile solutes make it harder for solvent to escape to vapor phase.