CHM2045 Lecture Notes - Lecture 10: Enthalpy, Exothermic Process, Thermodynamics
Document Summary
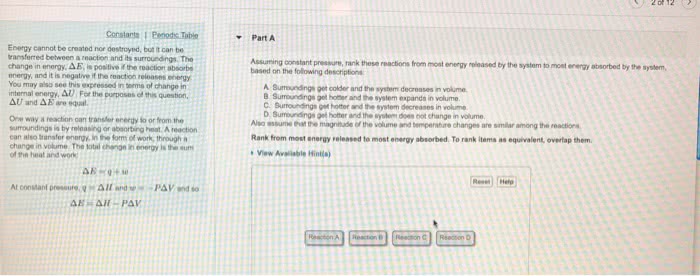
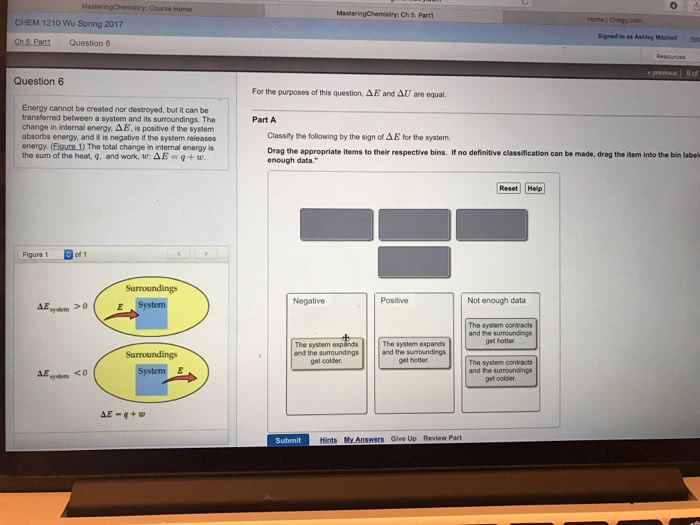
Heat in a reaction: endothermic- system that absorbs heat(cold, exothermic- system that loses heat(hot) Change in energy: a state function: a state function doesn"t depend on the path that was taken for the reaction to occur just on the difference between the two states. Altitude change between new orleans and denver. System vs. surroundings: system- what we are studying. Open- energy can be exchanged into the surroundings. Closed- energy can not be exchanged into the surroundings: surroundings- everything else, transferring energy: It is dif cult to calculate e for a given system but we can calculate the change in e: change in e= e( nal) - e(initial, change in e has: Chemical changes are present all around us under constant pressure, enthalpy measures the heat ow that surrounds us. H= e+pv: h= enthalpy, heat ow, e= internal energy, p= pressure, v= volume. Pressure volume work: the work for the expansion of gas, w= (-p)(change in volume, +change in volume: -w.