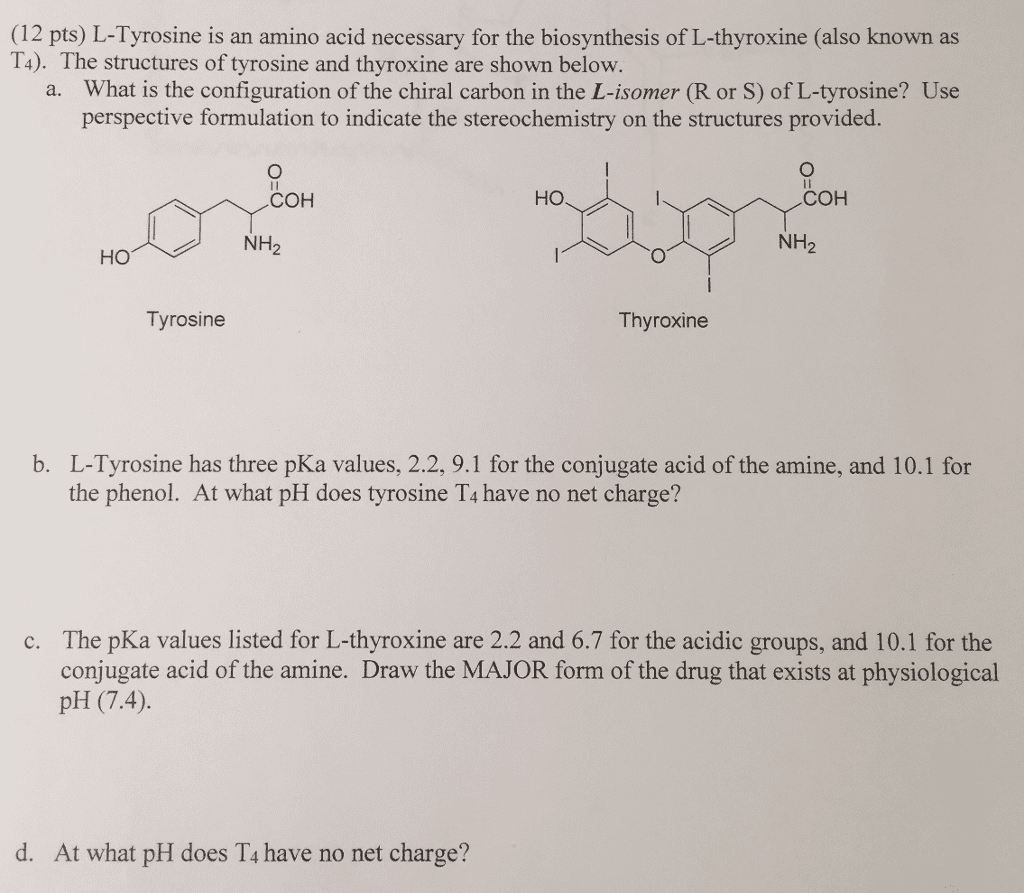
Options for Above:
Fumeric acid is
A. Smaller
B. Can Pack better in a lattice
C. Is more polar
D. Is larger
Maleic acid is
A. Larger
B. Smaller
C. More Polar
D. Less Polar
In maleic acid
A. Increased aromaticity
B. Intramolecular H-Bonding
C. A resonance structure
When removing he second H, pKa2 is lower for fumaric acid than maleic acid becase:
A. The two negatve charges in fumaric acid are farther apart
B. Fumaric acid has intramolecular hydrogen bonding
C. The two negatve charges in maleic acid are farther apart
D. Maleic acid is stabilized by resonance structures
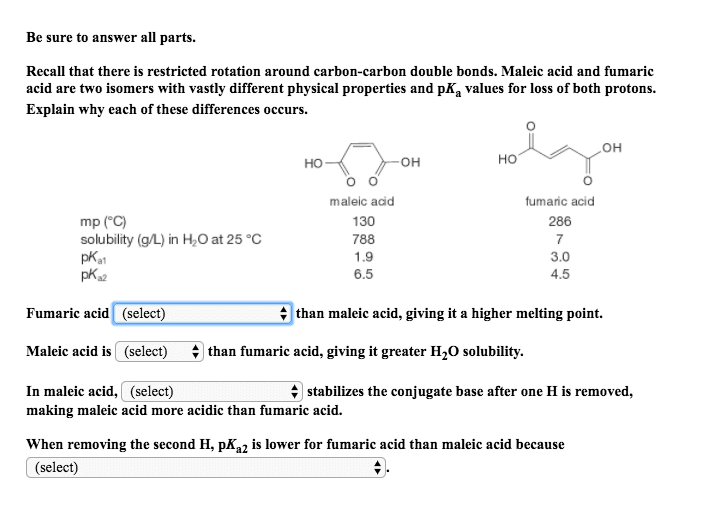
Be sure to answer all parts. Recall that there is restricted rotation around carbon-carbon double bonds. Maleic acid and fumaric acid are two isomers with vastly different physical properties and pKa values for loss of both protons. Explain why each of these differences occurs. OH HO OH HO o O maleic adid 130 788 1.9 6.5 fumaric acid 286 7 3.0 4.5 mp (C) solubility (g/L) in HO at 25 °C a1 a2 4 than maleic acid, giving it a higher melting point. Fumaric acid (select) Maleic acid is (select) In maleic acid, (select) Vthan fumaric acid, giving it greater H20 solubility 4 stabilizes the conjugate base after one H is removed, making maleic acid more acidic than fumaric acid. When removing the second H, pKa2 is lower for fumaric acid than maleic acid because (select)