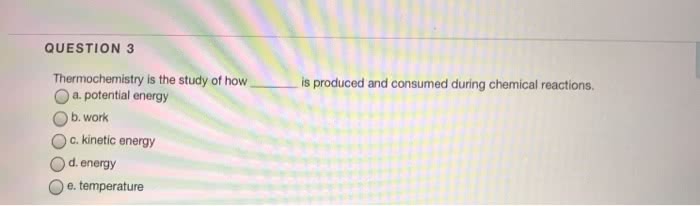
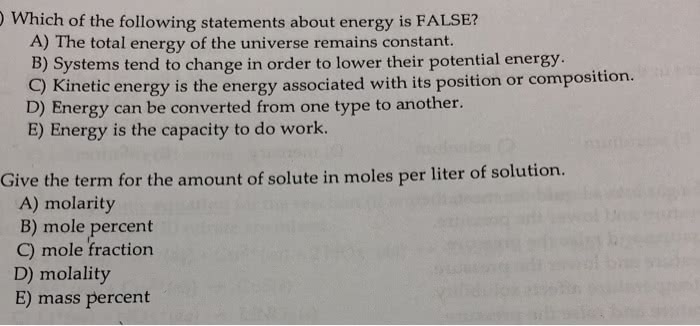CHEM-1601 Chapter Notes - Chapter 9: Thermochemistry, Thermodynamic System, Thermal Energy
Document Summary
List some examples of each: energy: the capacity to do work i. Think of energy as something that an object or set of objects possesses: work(w): the result of a force acting through a distance i. ii. List some examples of each: kinetic energy: the energy associated with the motion of an object i. A type of kinetic energy: thermal energy: associated with the temperature of an object, potential energy: the energy associated with the position or composition of an object i. How does it relate to energy exchanges between a thermodynamic system and its surroundings: law of conservation of energy: states that energy can be neither created nor destroyed i. Is internal energy a state function? a. b. Internal energy (e): the sum of the kinetic and potential energies of all of the particles that compose the system. If energy ows out of a chemical system and into the surroundings, what is the sign of esystem: negative.