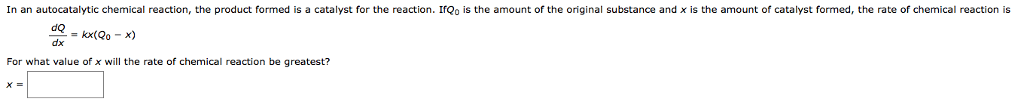1
answer
0
watching
209
views
9 Jun 2020
In a chemical reaction, the concentration of reactant is decreasing exponentiallly as reaction proceeds. The phase line for same is given as,

[X] represents the concentration of reaction. What is the maximum amount of reactant concetration that can be added for stable reaction and complete conversion of reactant to product ?
In a chemical reaction, the concentration of reactant is decreasing exponentiallly as reaction proceeds. The phase line for same is given as,
[X] represents the concentration of reaction. What is the maximum amount of reactant concetration that can be added for stable reaction and complete conversion of reactant to product ?
24 Jul 2020