4
answers
0
watching
1,549
views
12 Nov 2017
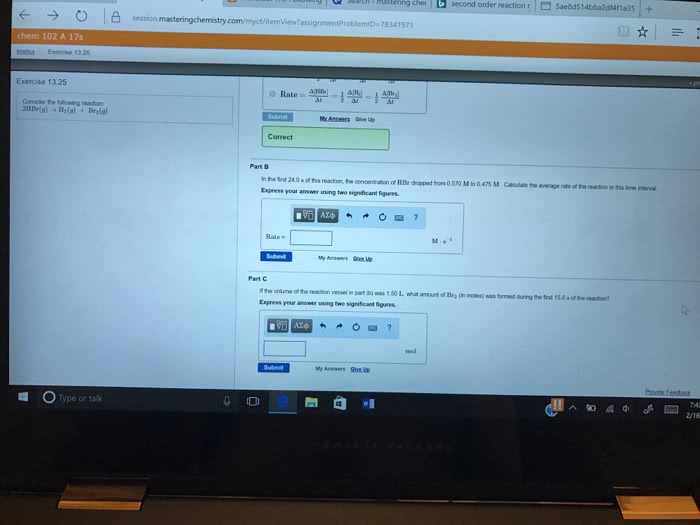
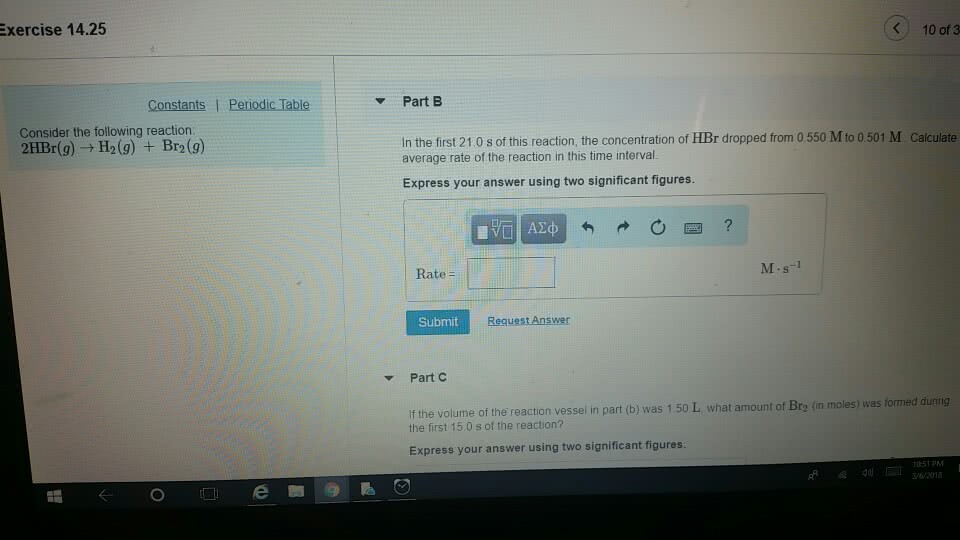
25. Consider the reaction: 2 HBr(8) — H2(g) + Brz(8) a. Express the rate of the reaction in terms of the change in concentration of each of the reactants and products. b. In the first 25.0 s of this reaction, the concentration of HBr dropped from 0.600 M to 0.512 M. Calculate the average rate of the reaction during this time interval. c. If the volume of the reaction vessel in part b was 1.50 L, what amount of Brz (in moles) was formed during the first 15.0 s of the reaction?
25. Consider the reaction: 2 HBr(8) — H2(g) + Brz(8) a. Express the rate of the reaction in terms of the change in concentration of each of the reactants and products. b. In the first 25.0 s of this reaction, the concentration of HBr dropped from 0.600 M to 0.512 M. Calculate the average rate of the reaction during this time interval. c. If the volume of the reaction vessel in part b was 1.50 L, what amount of Brz (in moles) was formed during the first 15.0 s of the reaction?
16 Jun 2023
Read by 1 person
Lelia LubowitzLv2
15 Nov 2017
Already have an account? Log in