4
answers
0
watching
375
views
30 Oct 2017
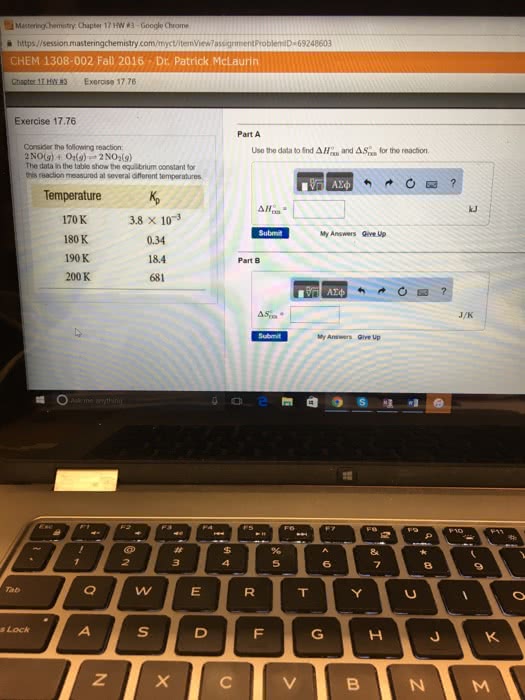
80. Consider the reaction: 2 NO(g) + O2(g) = 2 NO2(8) The following data show the equilibrium constant for this reac- tion measured at several different temperatures. Use the data to find A Hexn and ASixn for the reaction. Temperature 170 K. 3.8 x 10-3 180 K 0.34 190 K 18.4 200 K 681
80. Consider the reaction: 2 NO(g) + O2(g) = 2 NO2(8) The following data show the equilibrium constant for this reac- tion measured at several different temperatures. Use the data to find A Hexn and ASixn for the reaction. Temperature 170 K. 3.8 x 10-3 180 K 0.34 190 K 18.4 200 K 681
16 Jun 2023
Already have an account? Log in
Hubert KochLv2
31 Oct 2017
Already have an account? Log in