1
answer
0
watching
1,410
views
browncrow825Lv1
15 May 2019
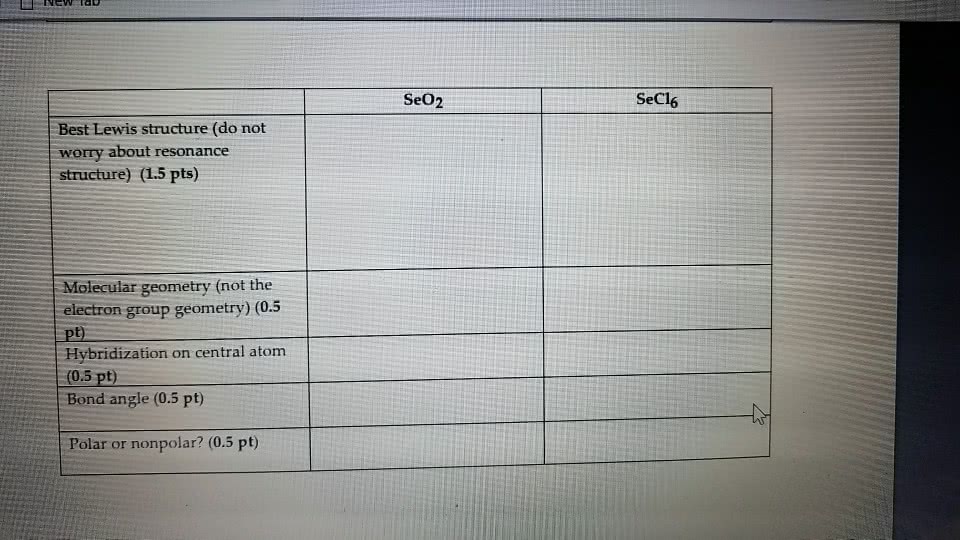
How do you draw the Lewis structure for SeO2. What is the electron geometry around the central atom?
How do you draw the Lewis structure for SeO2. What is the electron geometry around the central atom?
Bunny GreenfelderLv2
15 May 2019