1
answer
0
watching
185
views
11 Nov 2019
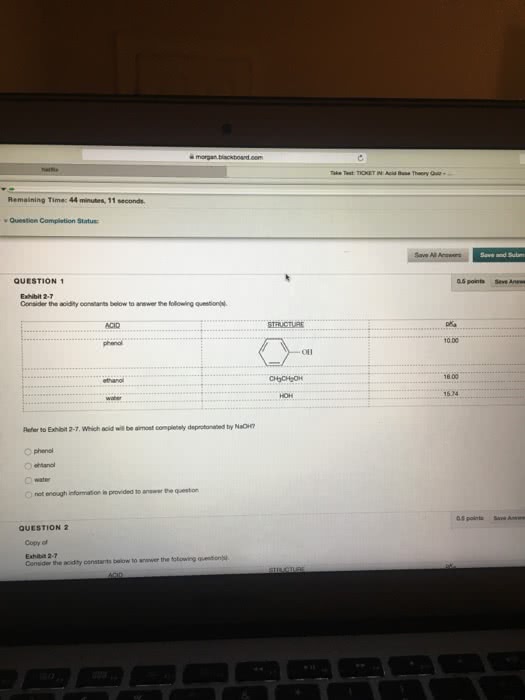

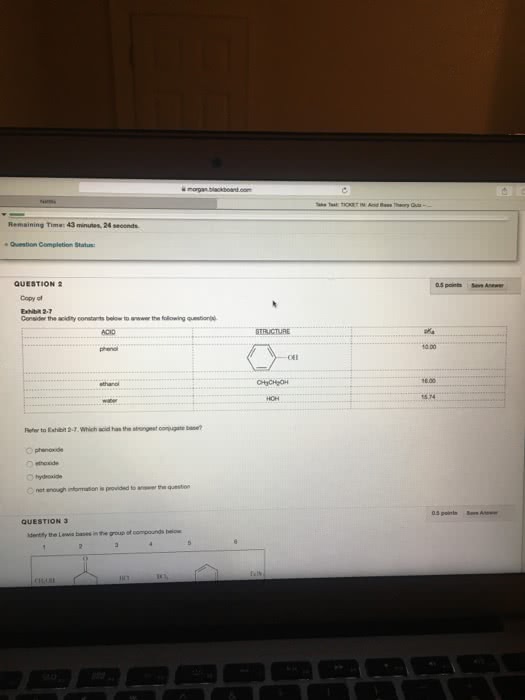
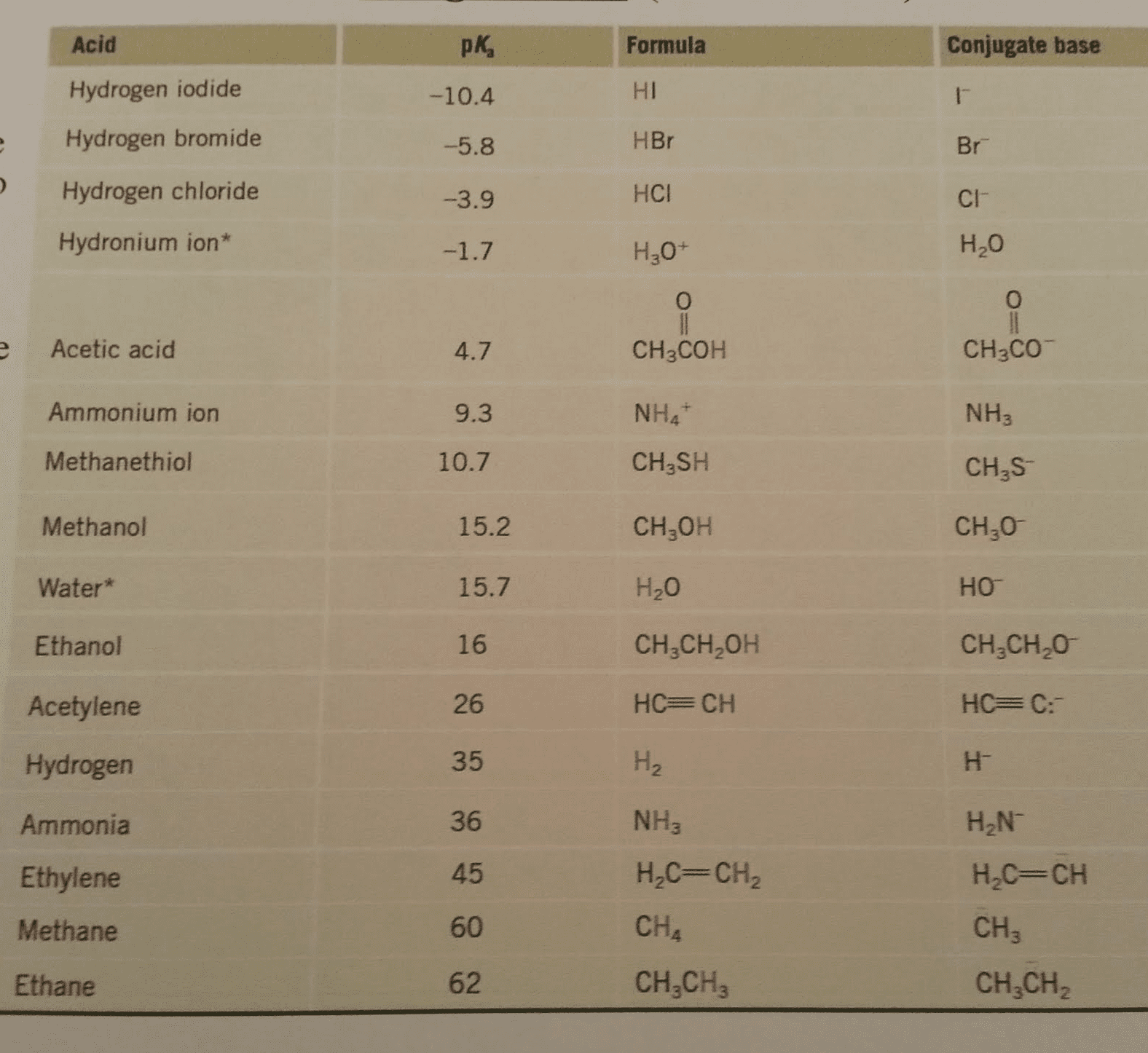
Consider the acidity constants below to answer the following question(s). Which acid will be almost completely deprotonated by NaOH? phenol ethanol water not enough information is provided to answer the question. Consider the acidity constants below to answer the following question(s).
1
answer
0
watching
185
views
For unlimited access to Homework Help, a Homework+ subscription is required.
Trinidad TremblayLv2
21 Sep 2019
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232
Related questions
Pyridine can be modified by attaching different groups on the asterisk. As a result of this, some properties of the compound may vary. One property that is affected is the acidity of the conjugate bases. The acid dissociation constants of the conjugate acids are given in the table below.

| Group Substituted in Asterisk | Ka of Conjugate Acid |
| NO2 |
 |
| Cl |
 |
| H |
 |
| CH3 |
 |
Suppose each conjugate acid is dissolved in water to yield a 0.050 M solution. Which solution would have the highest pH?