3
answers
0
watching
150
views
28 Sep 2019
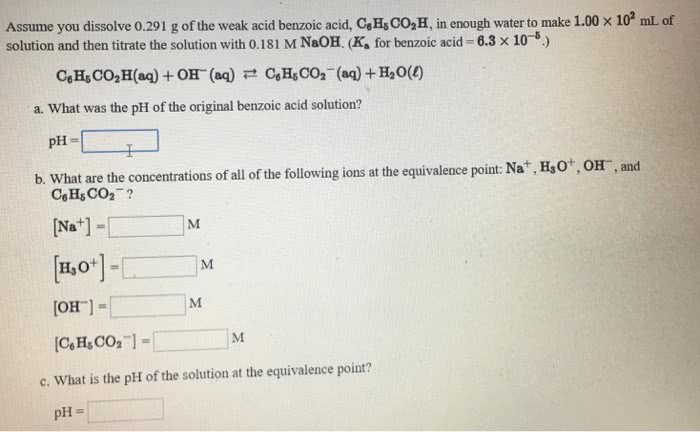
Assume you dissolve 0.253 g of the weak acid, benzoic acid (C6H5COOH) (HOBz), in enough water to make 100. mL, and then titrate the solution with 0.124 M NaOH(a) What was the original pH of the benzoic acid solution? (b) At the equivalence point what are the concentrations of [Na+] M ..... [OH-] M ..... [H+] ..... [BzO-] M(c) What is the pH at the equivalence point?
Assume you dissolve 0.253 g of the weak acid, benzoic acid (C6H5COOH) (HOBz), in enough water to make 100. mL, and then titrate the solution with 0.124 M NaOH(a) What was the original pH of the benzoic acid solution? (b) At the equivalence point what are the concentrations of [Na+] M ..... [OH-] M ..... [H+] ..... [BzO-] M(c) What is the pH at the equivalence point?
papayaprofessorLv10
12 Oct 2022
9 Oct 2022
Already have an account? Log in
Jean KeelingLv2
28 Sep 2019
Already have an account? Log in