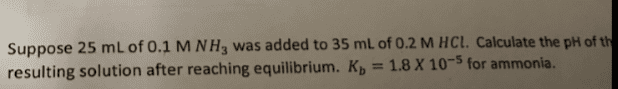
3
answers
0
watching
52
views
29 Sep 2019
URGENT QUESTION!!! Please answer this question with detailed explanation. Thank you.
A solution is made by mixing 100mL of 0.1 M HCl with 100 mL of 0.1 M NH3 at 25C (degrees celcius). What is the pH of the final solution? (Kb for NH3 = 1.8 x 10-5)
URGENT QUESTION!!! Please answer this question with detailed explanation. Thank you.
A solution is made by mixing 100mL of 0.1 M HCl with 100 mL of 0.1 M NH3 at 25C (degrees celcius). What is the pH of the final solution? (Kb for NH3 = 1.8 x 10-5)
1 Mar 2023
Already have an account? Log in
Beverley SmithLv2
29 Sep 2019
Already have an account? Log in