1
answer
0
watching
197
views
29 Nov 2019
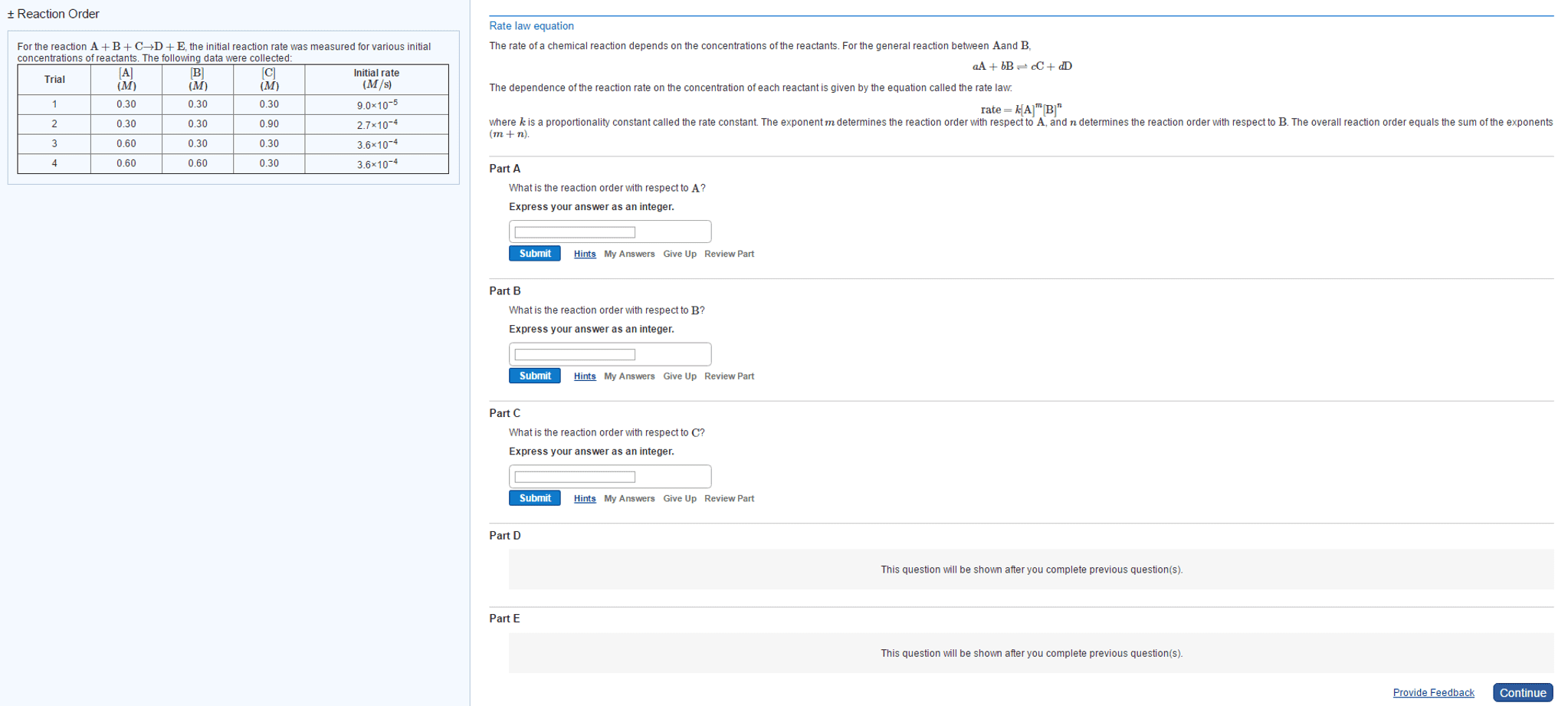
The rate of a chemical reaction depends on the concentrations ofthe reactants. For the general reaction between Aand B,
aA+bB?cC+dD
The dependence of the reaction rate on the concentration of eachreactant is given by the equation called the rate law:
rate=k[A]m[B]n
where k is a proportionality constant called the rateconstant. The exponent m determines the reaction orderwith respect to A, and n determines the reaction orderwith respect to B. The overall reaction order equals the sum of theexponents (m+n).
For the reaction A+B+C?D+E, the initial reaction rate wasmeasured for various initial concentrations of reactants. Thefollowing data were collected:
Trial [A]
(M) [B]
(M) [C]
(M) Initial rate
(M/s) 1 0.40 0.40 0.40 1.2
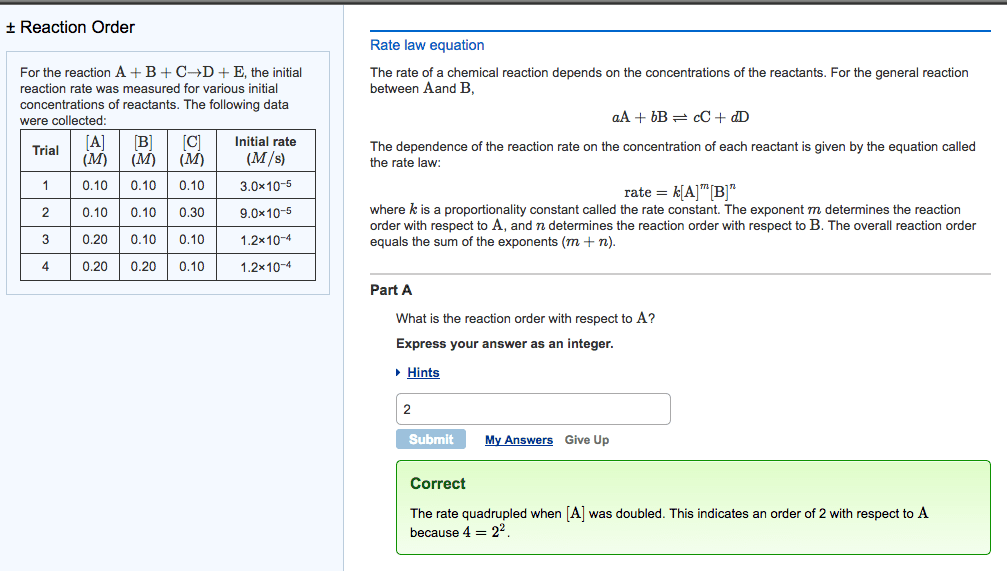
The rate of a chemical reaction depends on the concentrations ofthe reactants. For the general reaction between Aand B,
aA+bB?cC+dD
The dependence of the reaction rate on the concentration of eachreactant is given by the equation called the rate law:
rate=k[A]m[B]n
where k is a proportionality constant called the rateconstant. The exponent m determines the reaction orderwith respect to A, and n determines the reaction orderwith respect to B. The overall reaction order equals the sum of theexponents (m+n).
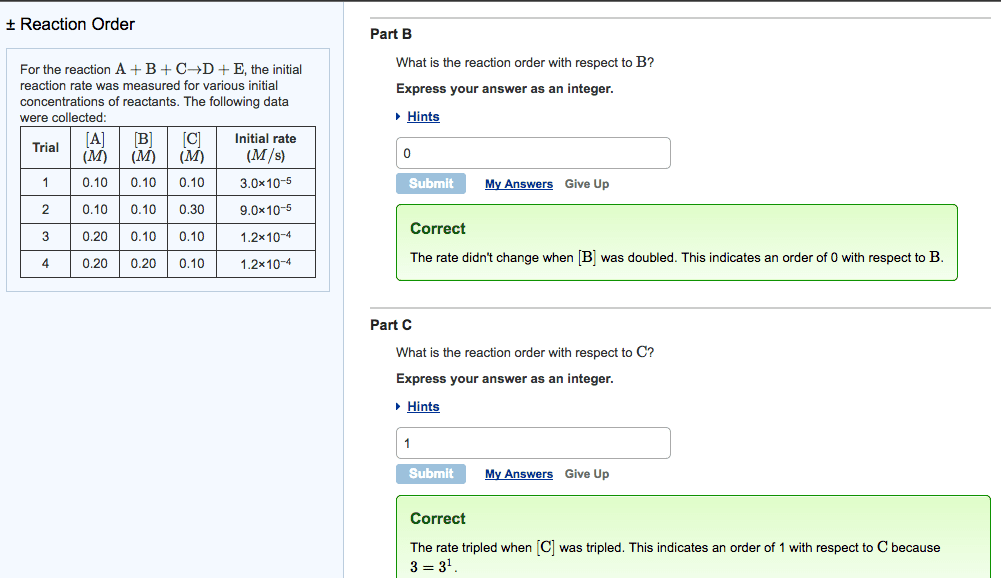
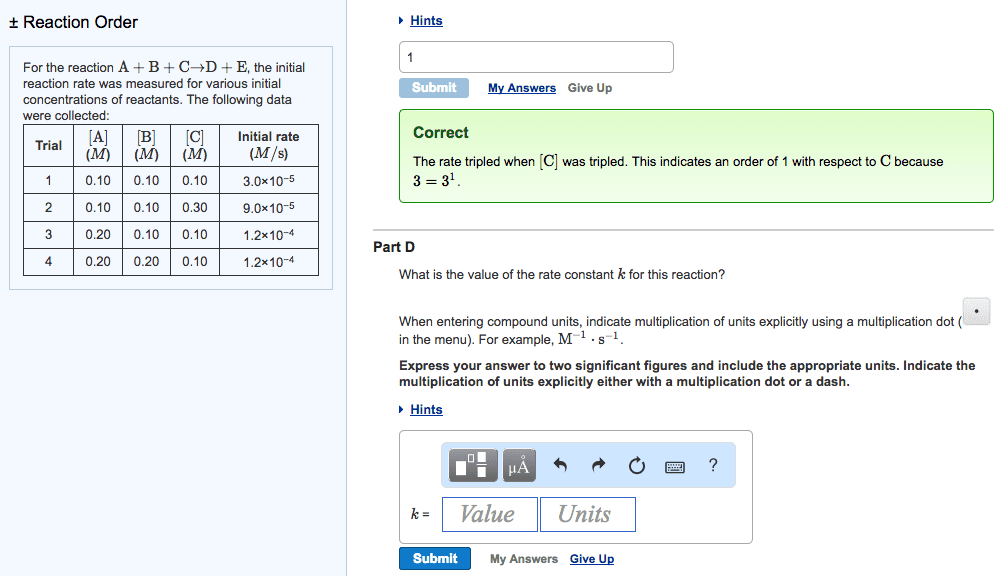
For the reaction A+B+C?D+E, the initial reaction rate wasmeasured for various initial concentrations of reactants. Thefollowing data were collected:
| Trial | [A] (M) | [B] (M) | [C] (M) | Initial rate (M/s) |
| 1 | 0.40 | 0.40 | 0.40 | 1.2 |
Irving HeathcoteLv2
17 Dec 2019