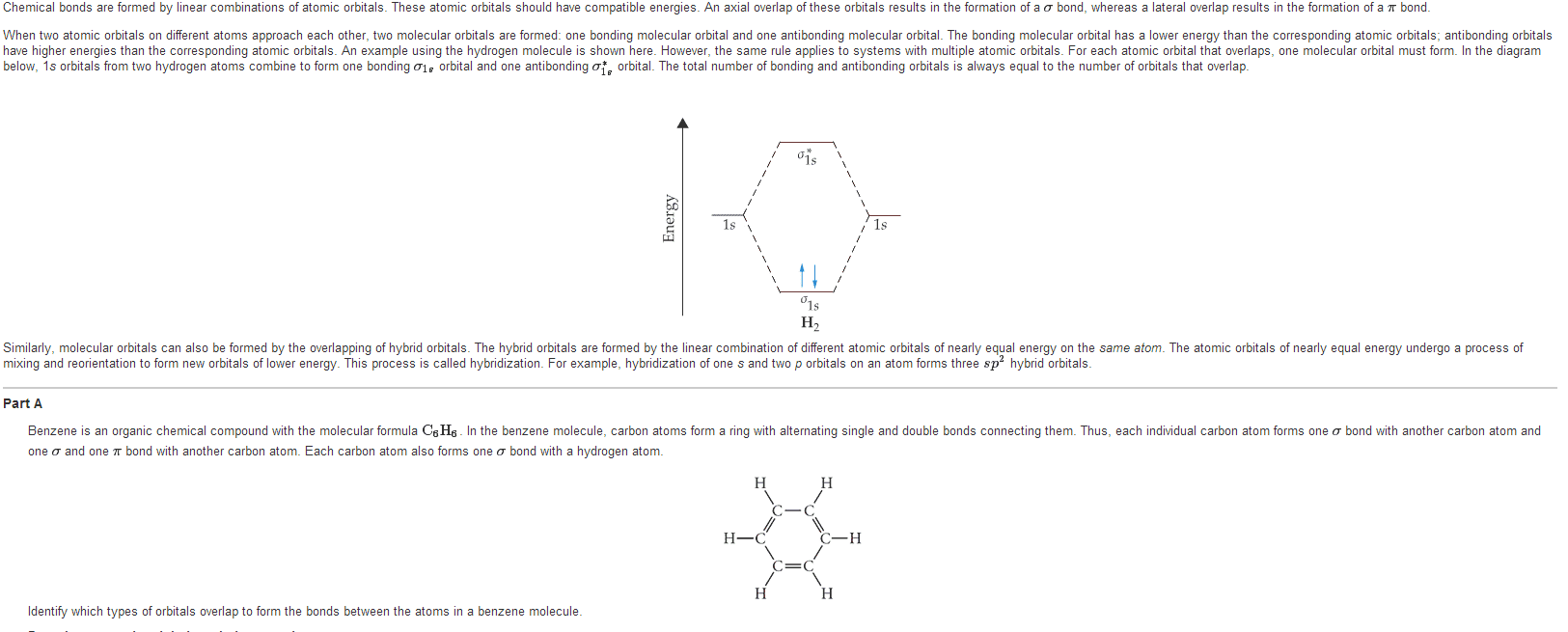
1
answer
0
watching
109
views
13 Dec 2019
Which of the following statements is incorrect regardingmolecular orbital theory?
A. the combination of two atomic orbitals produces twomolecular orbitals. B. the bond order is calculated in the same way as withLewis structures. C. molecules with an even number of electrons will havebond orders which are whole numbers D. combination of two 2p atomic orbitals maygive rise to either sigma(?) or pi(?) type molecular orbitals E. all diatomic molecules with an odd number ofelectrons will be paramagnetic
***MUST SHOW ALL WORK
Which of the following statements is incorrect regardingmolecular orbital theory?
| A. | the combination of two atomic orbitals produces twomolecular orbitals. | |
| B. | the bond order is calculated in the same way as withLewis structures. | |
| C. | molecules with an even number of electrons will havebond orders which are whole numbers | |
| D. | combination of two 2p atomic orbitals maygive rise to either sigma(?) or pi(?) type molecular orbitals | |
| E. | all diatomic molecules with an odd number ofelectrons will be paramagnetic ***MUST SHOW ALL WORK |
1
answer
0
watching
109
views
For unlimited access to Homework Help, a Homework+ subscription is required.
Lelia LubowitzLv2
17 Dec 2019
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232