Ethyl ether (CAS# 60-29-7; Formula C2H5OC2H5) is a very useful chemical that is found in many laboratories. It is a Class IA Flammable Liquid, with an OSHA PEL of 400 ppm, a flammable range of 1.9% to 36%, and an auto-ignition temperature of 320 F (160 C). Because it is a good solvent, evaporates easily, and is only slightly soluble in cold water, you decide to use it as an extraction solvent in a separatory funnel. After the extraction, you plan to rapidly evaporate the ether inside a fume hood using an electric hot-plate that is set at 318 degrees F. Unfortunately, while preparing your extraction process you dropped the bottle of ether and it broke on the floor. NO ONE is injured, but you now have a large mess to clean up. Your lab is located in a small town and you are the only member of the local volunteer Haz-Mat response team. This means you have to take care of the mess yourself - you can't just leave the lab and call 911 because you ARE 911. Before you begin the cleanup, you determine that in the area of the spill the concentration of ether vapors is 2000 ppm and the oxygen concentration is 19.3%. The most appropriate PPE to wear during the cleanup is:
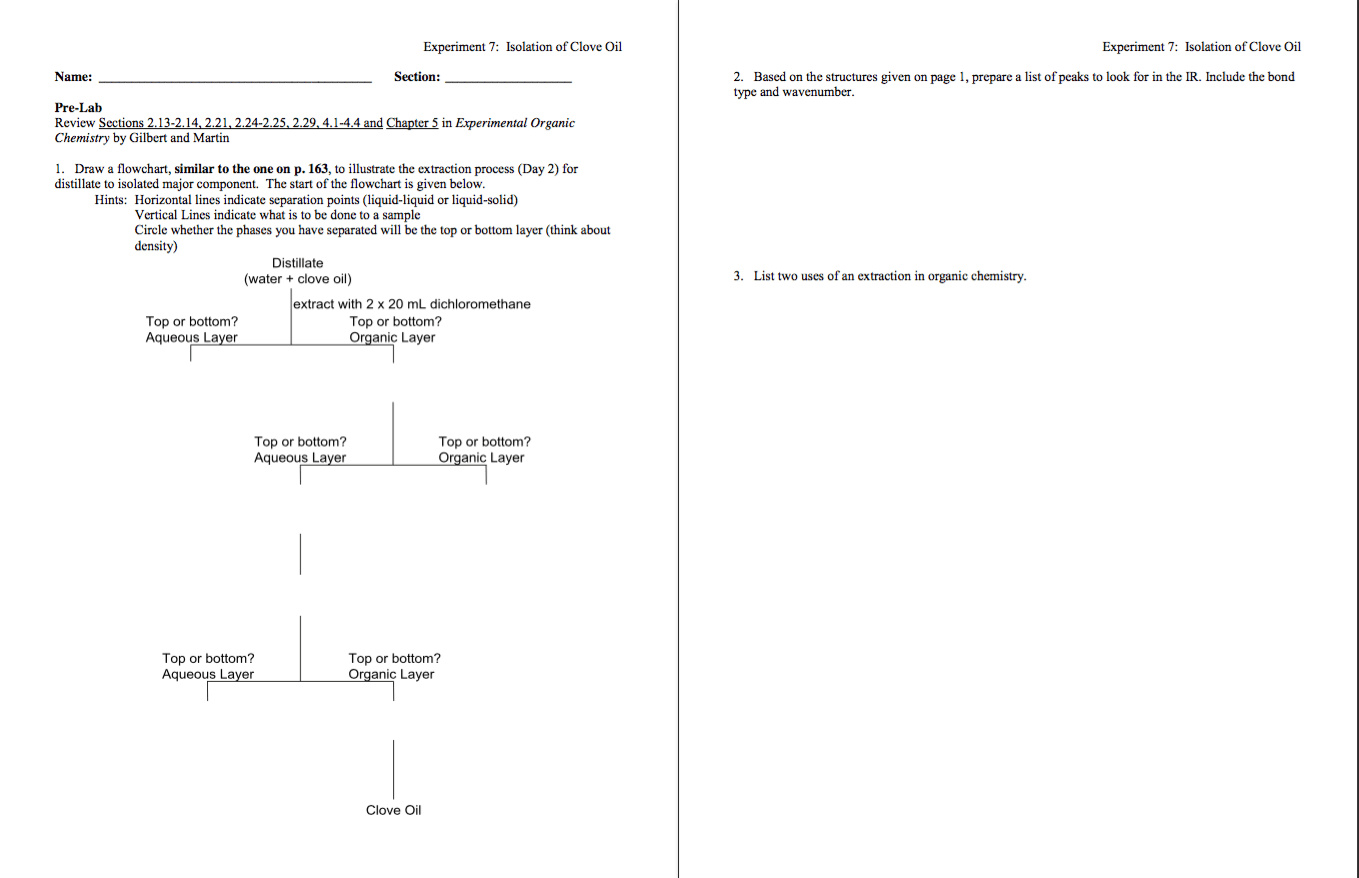
Level D (gloves, lab coat, eye protection, long pants, and closed shoes). No respiratory protection is necessary because the spill will be cleaned up long-before your 8-hour Time Weighted Average exposure reaches 400 ppm, and OSHA does not have a STEL for ether. Fire isn't an immediate concern because ether vapors are heavier than air and the spill is on the floor while the hot plate is several feet above in a fume hood.
Level C. Use an air-purifying respirator with "organic vapor" cartridges installed that are capable of reducing the ether concentration that you breathe to less than 100 ppm. Also wear solvent-resistant gloves and a chemical suit. Before you start to clean up the spill, you will turn off the hot plate.
Level B. Use a Self-Contained Breathing Apparatus (SCBA) because of the relatively high vapor concentration and the relatively low oxygen concentration. Although you will wear appropriate gloves and a chemical suit, there is no need for an encapsulated (level A) suit because your skin does not need to be completely protected from the vapors like your lungs need to be protected.
Level A. Top-of-the-line, first-class protection. SCBA with fully-encapsulated, chemical-resistant suit and gloves. As soon as you dropped the bottle, you shut off the hot plate and left the area to go put on your enhanced PPE (because you are smart, you were already wearing level "D" with all-cotton clothes, leather shoes, and a fire-resistant lab coat). You will use tools while cleaning the broken glass so that you do not accidentally cut a hole in your very expensive Level-A suit.None of the above. In the box below, briefly explain why options (A) through (D) are not entirely correct and what you believe is a better way to manage this spill.
Explain your answer:
Ethyl ether (CAS# 60-29-7; Formula C2H5OC2H5) is a very useful chemical that is found in many laboratories. It is a Class IA Flammable Liquid, with an OSHA PEL of 400 ppm, a flammable range of 1.9% to 36%, and an auto-ignition temperature of 320 F (160 C). Because it is a good solvent, evaporates easily, and is only slightly soluble in cold water, you decide to use it as an extraction solvent in a separatory funnel. After the extraction, you plan to rapidly evaporate the ether inside a fume hood using an electric hot-plate that is set at 318 degrees F. Unfortunately, while preparing your extraction process you dropped the bottle of ether and it broke on the floor. NO ONE is injured, but you now have a large mess to clean up. Your lab is located in a small town and you are the only member of the local volunteer Haz-Mat response team. This means you have to take care of the mess yourself - you can't just leave the lab and call 911 because you ARE 911. Before you begin the cleanup, you determine that in the area of the spill the concentration of ether vapors is 2000 ppm and the oxygen concentration is 19.3%. The most appropriate PPE to wear during the cleanup is:
Level D (gloves, lab coat, eye protection, long pants, and closed shoes). No respiratory protection is necessary because the spill will be cleaned up long-before your 8-hour Time Weighted Average exposure reaches 400 ppm, and OSHA does not have a STEL for ether. Fire isn't an immediate concern because ether vapors are heavier than air and the spill is on the floor while the hot plate is several feet above in a fume hood.
Level C. Use an air-purifying respirator with "organic vapor" cartridges installed that are capable of reducing the ether concentration that you breathe to less than 100 ppm. Also wear solvent-resistant gloves and a chemical suit. Before you start to clean up the spill, you will turn off the hot plate.
Level B. Use a Self-Contained Breathing Apparatus (SCBA) because of the relatively high vapor concentration and the relatively low oxygen concentration. Although you will wear appropriate gloves and a chemical suit, there is no need for an encapsulated (level A) suit because your skin does not need to be completely protected from the vapors like your lungs need to be protected.
Level A. Top-of-the-line, first-class protection. SCBA with fully-encapsulated, chemical-resistant suit and gloves. As soon as you dropped the bottle, you shut off the hot plate and left the area to go put on your enhanced PPE (because you are smart, you were already wearing level "D" with all-cotton clothes, leather shoes, and a fire-resistant lab coat). You will use tools while cleaning the broken glass so that you do not accidentally cut a hole in your very expensive Level-A suit.None of the above. In the box below, briefly explain why options (A) through (D) are not entirely correct and what you believe is a better way to manage this spill.
Explain your answer: