1
answer
0
watching
214
views
13 Dec 2019
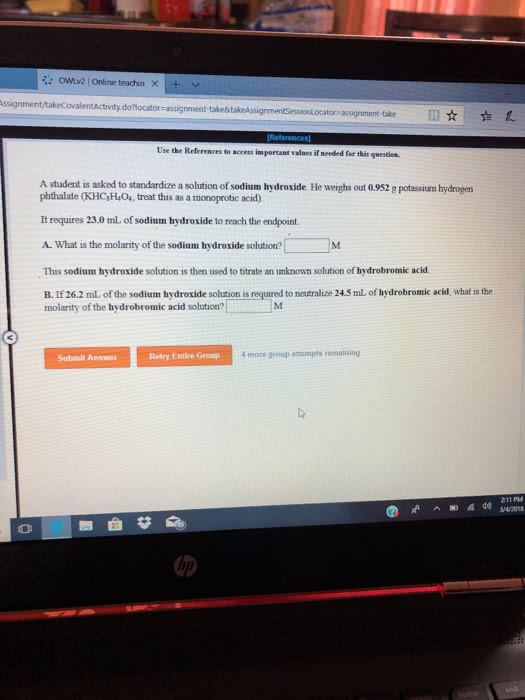
A 13.1 g sample of an aqueous solution of hydrobromic acid contains an unknown amount of the acid.
If 21.3 mL of 1.99 M sodium hydroxide are required to neutralize the hydrobromic acid, what is the percent by mass of hydrobromic acid in the mixture?
? % by mass
In the laboratory a student combines 39.3 mL of a 0.381 M nickel(II) fluoride solution with 17.6 mL of a 0.470 M ammonium fluoridesolution.
What is the final concentration of fluoride anion ?
M
Write a net ionic equation for the reaction that occurs when aqueous solutions of acetic acid and ammonia are combined.
? + ? ? + ? (aq)(s)(l)(g)
A 13.1 g sample of an aqueous solution of hydrobromic acid contains an unknown amount of the acid.
If 21.3 mL of 1.99 M sodium hydroxide are required to neutralize the hydrobromic acid, what is the percent by mass of hydrobromic acid in the mixture?
? % by mass
In the laboratory a student combines 39.3 mL of a 0.381 M nickel(II) fluoride solution with 17.6 mL of a 0.470 M ammonium fluoridesolution.
What is the final concentration of fluoride anion ?
M
Write a net ionic equation for the reaction that occurs when aqueous solutions of acetic acid and ammonia are combined.
| ? | + | ? | ? | + | ? (aq)(s)(l)(g) |
|---|
1
answer
0
watching
214
views
For unlimited access to Homework Help, a Homework+ subscription is required.
Nelly StrackeLv2
17 Dec 2019
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232