1
answer
0
watching
213
views
31 May 2020
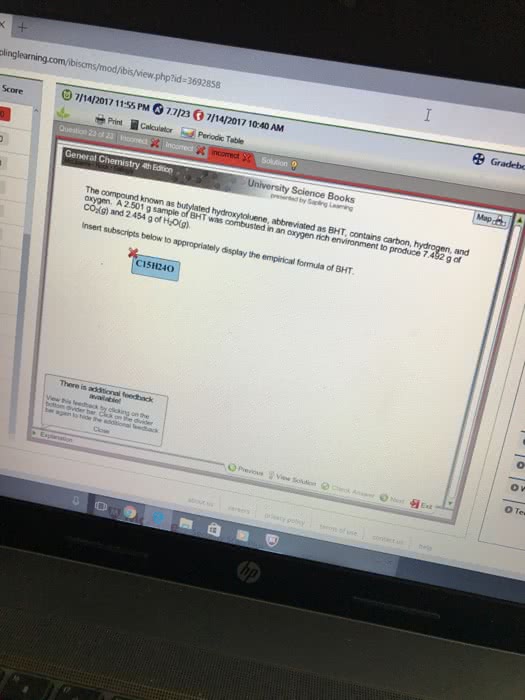
An unknown compound contains  ,
,  ,
,  and is combustion with excess oxygen.
and is combustion with excess oxygen.  of sample is combusted to form
of sample is combusted to form  of
of  and
and  of
of  . What is the empirical formula?
. What is the empirical formula?
An unknown compound contains ,
,
and is combustion with excess oxygen.
of sample is combusted to form
of
and
of
. What is the empirical formula?
Tod ThielLv2
24 Jul 2020