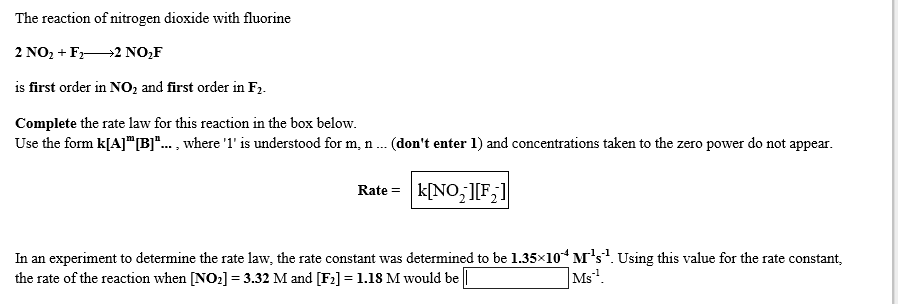1
answer
0
watching
190
views
16 Jun 2020
Determine the unit of  , the rate constant in the rate law of a second-order chemical reaction:
, the rate constant in the rate law of a second-order chemical reaction:

where the unit of rate is  ,
,  is
is  .
.
Determine the unit of , the rate constant in the rate law of a second-order chemical reaction:
where the unit of rate is ,
is
.
1
answer
0
watching
190
views
For unlimited access to Homework Help, a Homework+ subscription is required.
24 Jul 2020
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232
Related questions
Some measurements of the initial rate of a certain reaction are given in the table below.
| N2 | H2 | initial rate of reaction |
|---|---|---|
| 2.49M | 0.760M | 91.0/Ms |
| 1.15M | 0.760M | 19.4/Ms |
| 2.49M | 3.02M | 362./Ms |
Use this information to write a rate law for this reaction, and calculate the value of the rate constant k
Round your value for the rate constant to 2 significant digits. Also be sure your answer has the correct unit symbol.
rate = k__________?
k = ___________?