1
answer
0
watching
165
views
27 Jun 2020
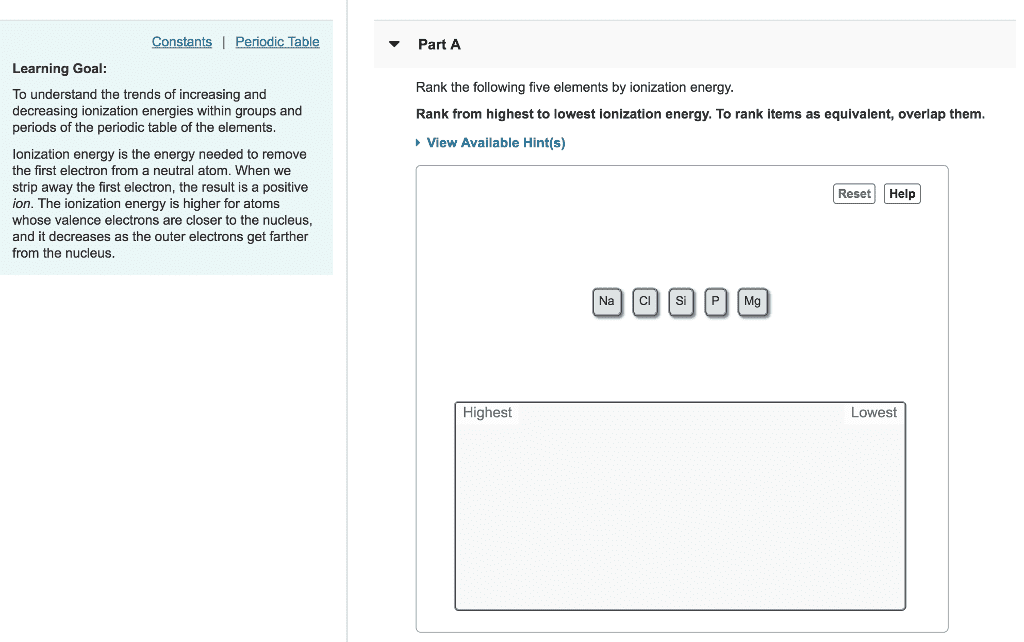
Which of the elements shows the least value of ionization energy within their periods?
Which of the elements shows the least value of ionization energy within their periods?
1
answer
0
watching
165
views
For unlimited access to Homework Help, a Homework+ subscription is required.
1 Sep 2020
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232
Related questions
Part A
Which of the following elements has the greatest metallic character (which is more metallic)?
| P | ||
| Sb | ||
| In | ||
| Ag | ||
| Au |
Part B
For the element calcium, which of the following ionization energies should show a large jump in magnitude?
| first ionization energy | ||
| second ionization energy | ||
| third ionization energy | ||
| fourth ionization energy | ||
| fifth ionization energy | ||
| sixth ionization energy | ||
| seventh ionization energy |
Part C
Which of the following shows irregular behavior in it's electron configuration?
| Mg | ||
| Cr | ||
| Co | ||
| P | ||
| Ar |