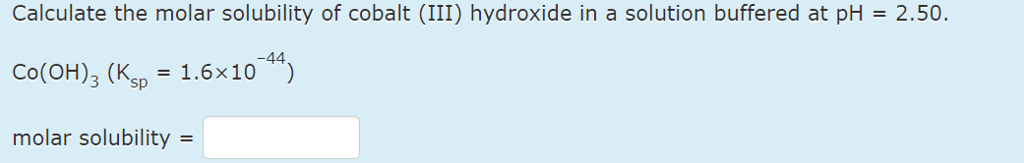1
answer
0
watching
378
views
30 Jun 2020
What is the molar solubility of Cr(OH)3 in a buffered solution at pH = 11? (Cr(OH)3 Ksp = 6.7 x 10-31)
What is the molar solubility of Cr(OH)3 in a buffered solution at pH = 11? (Cr(OH)3 Ksp = 6.7 x 10-31)
1 Sep 2020