CHEM 122 Chapter : 15.4 The Integrated Rate Law.docx
85 views2 pages
13 Dec 2012
School
Department
Course
Professor
Document Summary
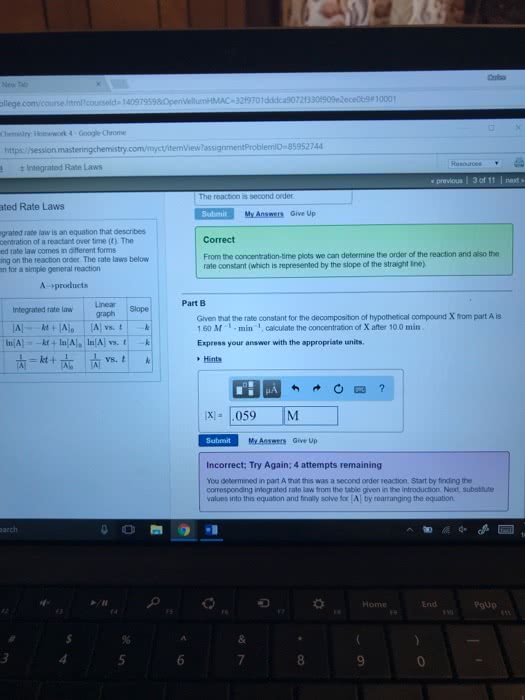
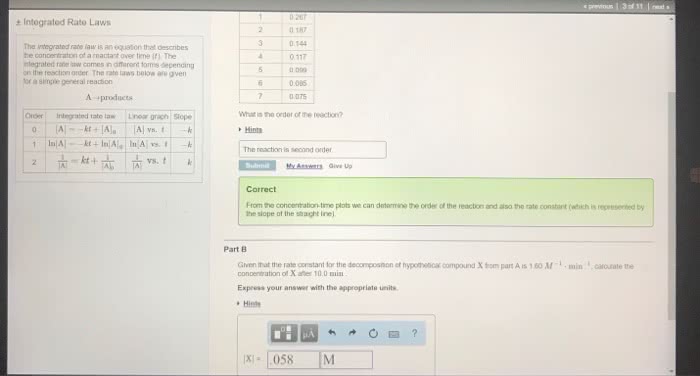
Expressing the rate as a function of time. Formed from the given differential rate law. If reaction is in first order, rate = -d[n2o5]/dt = k[n2o5] Integrated rate law: ln[n2o5] = -kt + ln[n2o5]o. The equation shows how [a] depends on time. If [a]o and value of the rate constant, k, are known, the concentration of a at any time can be calculated. Y = mx + b; y = ln[a] x = t m = -k b = ln[a]o. Reaction is in first order if a plot of ln[a] vs. time gives a straight line. The integrated rate law can also be represented in terms of the ratio of [a] and. Time required for a reactant to reach half its original concentration t1/2 = 0. 693/k *half-life does not depend on initial concentration. If the reaction is second order, rate = -d[a]/dt = k[a]2. Integrated rate law: 1/[a] = kt + 1/[a]o.
Get access
Grade+20% off
$8 USD/m$10 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
40 Verified Answers
Class+
$8 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
30 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232