CHEM 2OA3 Lecture : 07-03 - Lewis Structures, Resonance, Hybridization.docx
Document Summary
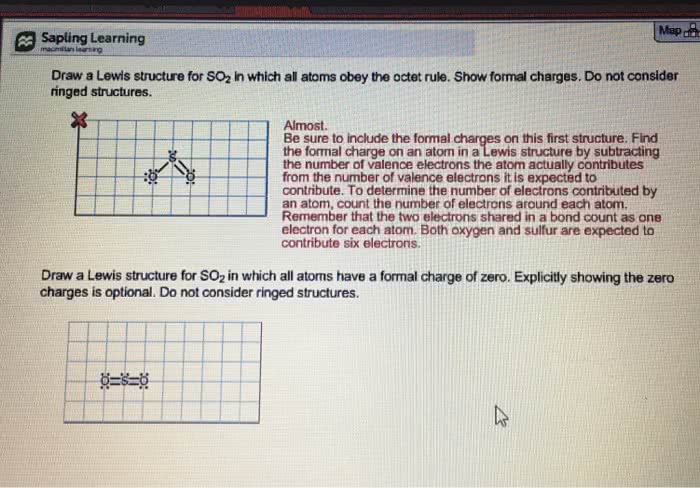
Lewis structures: draw molecular skeleton (given, count total # of valence electrons, octet (duet) rule (most important, wins over all other rules, provide octets (duets for hydrogen) around all atoms, take care of charges, if any. Charges occur when the formal effective electron count around the nucleus differs from valence electron count (from periodic table: effective electron count: each bond with two shared electrons counts as 1e; lone pairs count as 2e. Octet rule negative charge on more electronegative, positive charge on less electronegative minimize charge separation. When a molecule has multiple lewis structures that are valid, it has resonance (the structures are resonance structures ) Radical: molecule with single electron instead of an electron pair connected to it: radical doesn"t push as hard as an electron pair does, so it can skew the geometric shape s orbital ( ball )