If a propanoic acid/propanoate buffer is 0.035 M in propanoic acidand has a pH of 4.59, what is the molar concentration of thepropanoate ion? The pKa of propanoic acid is 4.89.
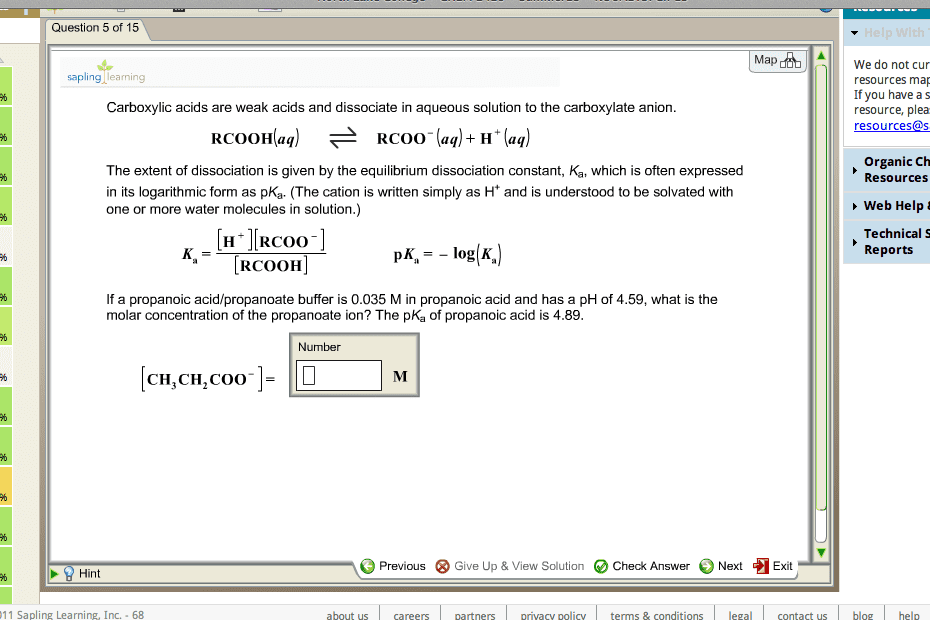
Carboxylic acids are weak acids and dissociate in aqueous solution to the RCOOH(aq) rightarrow RCOO-(aq) + H+(aq) The extent of dissociation is given by the equilibrium dissociation constant, Ka, which is often expressed in its logarithmic form as pKa. (The cation is written simply as H+ and is understood to be solvated with one or more water molecules in solution.) K1 = [h+][RCOO-] / [RCOOH] pkn = -log(Ka) If a propanoic acid/propanoate buffer is 0.035 M in propanoic acid and has a pH of 4.59, what is the molar concentration of the propanoate ion? The pKa of propanoic acid is 4.89. [CH3 CH2 COO-] =
Show transcribed image text Carboxylic acids are weak acids and dissociate in aqueous solution to the RCOOH(aq) rightarrow RCOO-(aq) + H+(aq) The extent of dissociation is given by the equilibrium dissociation constant, Ka, which is often expressed in its logarithmic form as pKa. (The cation is written simply as H+ and is understood to be solvated with one or more water molecules in solution.) K1 = [h+][RCOO-] / [RCOOH] pkn = -log(Ka) If a propanoic acid/propanoate buffer is 0.035 M in propanoic acid and has a pH of 4.59, what is the molar concentration of the propanoate ion? The pKa of propanoic acid is 4.89. [CH3 CH2 COO-] =






