CHEM 1AA3 Lecture Notes - Buffer Solution, Conjugate Acid, Acid Strength
120 views7 pages
19 Mar 2013
School
Department
Course
Professor
Document Summary
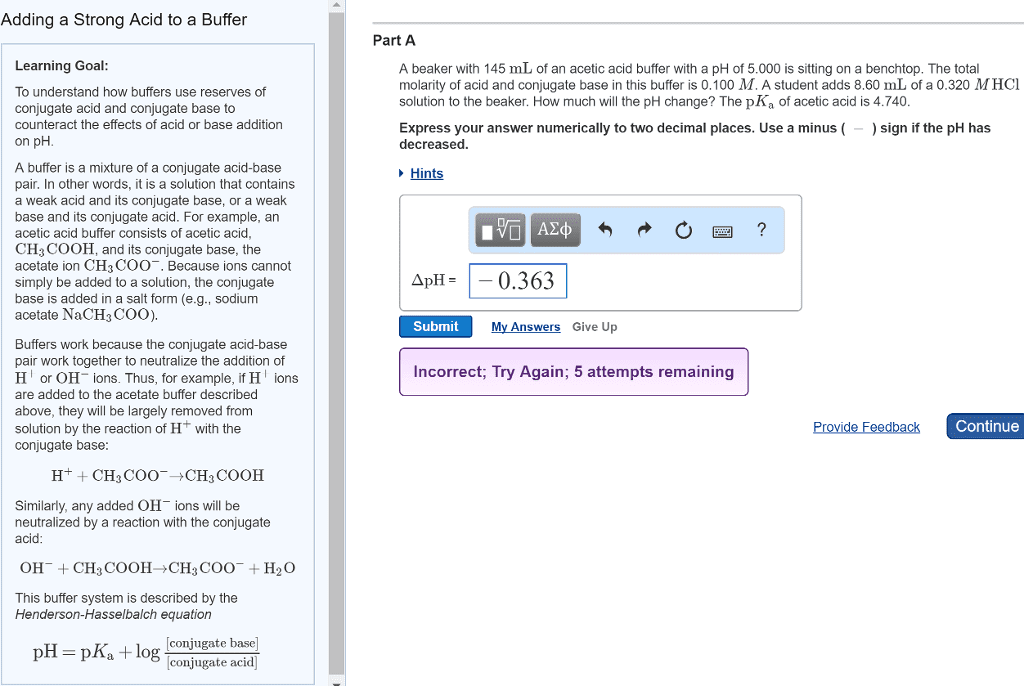
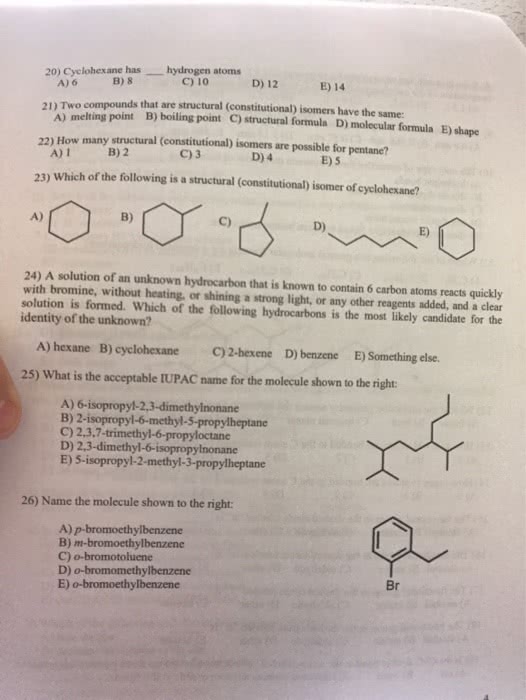
Bl acid h+ donor, bl base h+ acceptor. Strong acids/bases 100% ionization in water. Weak acids/bases equilibrium reaction with water (ka and kb values: weak acid ka < 1, pka > 1, weak base kb < 1, pkb > 1. Ph + poh = 14 = pka + pkb. Ph of salt acidic (nh4cl), neutral (libr), basic (nach3coo) A solution of a weak acid or weak base initially contains a second source of one of the ions produced in the ionization of the acid or base. The added ions are said to be common to the weak acid or base. Nach3coo na+ + ch3coo: when a salt supplies the common ion ch3coo-, the equilibrium shifts to form more ch3cooh. Solutions of weak bases and their salts: ex. + + cl: when a salt supplies the common anion nh4. +, the equilibrium shifts to form more nh3.
Get access
Grade+20% off
$8 USD/m$10 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
40 Verified Answers
Class+
$8 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
30 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232