Chemistry 1027A/B Lecture Notes - Alkene, Nucleophile, Ion
22 views1 pages
4 Nov 2013
School
Department
Course
Professor
Document Summary
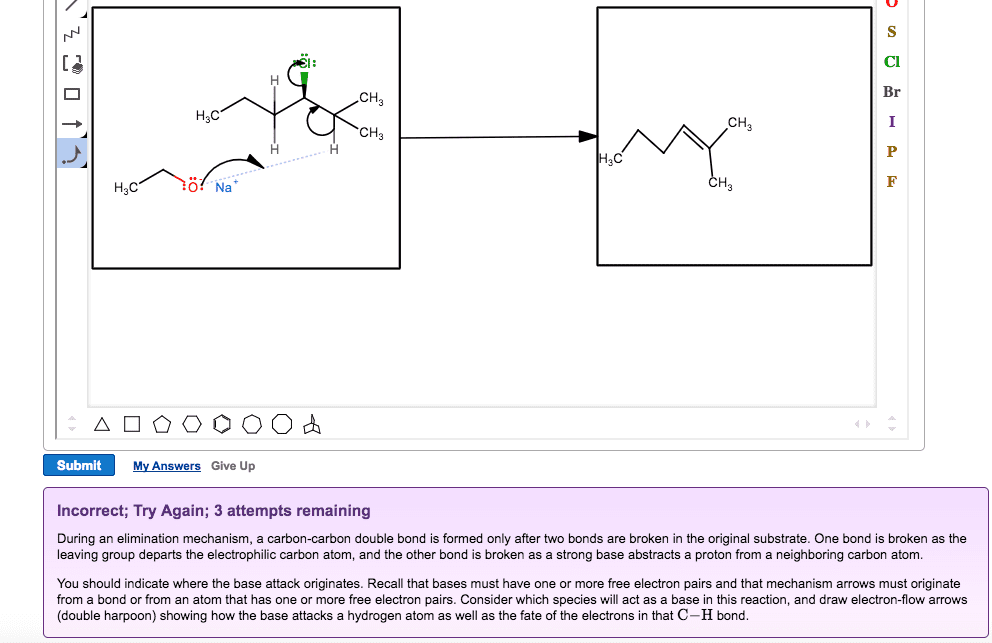
Key concepts in organic chemistry: haloalkanes part ii (usselman version) C-lg can undergo an elimination reaction, in addition to the substitution reactions already discussed. In elimination reactions, the nu: acts as a base (b) and attacks an h instead of attacking c as a nucleophile. Because the h atom and the lg come off c atoms that are adjacent (often labelled and ), this reaction is called a elimination. There are two molecular features which act to increase the yield of elimination products at the expense of substitution products: i) a large anion, and ii) a stable alkene as a product. By zaitsev"s rule, the alkene with the most alkyl groups will form in greater amounts. The e2 elimination mechanism is similar to the sn2 mechanism. Elimination products by an e2 mechanism often accompany substitution products by an sn2 mechanism. The e1 elimination mechanism is similar to the sn1 mechanism.
Get access
Grade+20% off
$8 USD/m$10 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
40 Verified Answers
Class+
$8 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
30 Verified Answers