CHM120H5 Lecture Notes - Crc Handbook Of Chemistry And Physics, Chromate And Dichromate, Boca Raton, Florida
Document Summary
Get access


Related Documents
Related Questions
Lab Report Assistant
This document is not meant to be a substitute for a formal laboratory report. The Lab Report Assistant is simply a summary of the experimentâs questions, diagrams if needed, and data tables that should be addressed in a formal lab report. The intent is to facilitate studentsâ writing of lab reports by providing this information in an editable file which can be sent to an instructor.
Exercise 1: Qualitative Anion Tests
Data Table 1. Anion Confirmation Tests.
| Chemical | Anion | Addition of AgNO3 | Addition of HCl |
| NaBr | Bromide | It became white and cloudy | Still was white and cloudy |
| Na2CO3 | Carbonate | Clear inside white and cloudy around the outside | Still white and cloudy but bubbles start to form |
| NaCl | Chloride | Turn white cloudy on bottom clear on top | Still white cloudy on bottom clear on top |
| NaI | Iodide | In the middle very dark and cloudy fades out lighter white around the middle | Became a little separate |
| Na2SO4 | Sulfate | Clear | Became white and cloudy |
| Na2S | Sulfide | Turn a brown color in the middle but separated from the clear | The brown becomes more separate from the clear |
| Unknown #103 | CO3^2- | White and cloudy Small particles | Bubbles form white and cloudy |
| Unknown #104 | S^2- | Turn brown color I the middle around clear | Some of the brown disappeared and become separate |
Flow chart:
Solution containing: Br-, CO3^2-, Cl-, I-, SO4^2-, and S^2-
{AgBr(cloudy), Ag2CO3(cloudy), AglCloudy),Agl(very dark), Ag2S-(brown)} {So4^2-}
{AgBr(cloudy), AgCl(cloudy), Agl(very cloudy), Ag2S(brown)} {CO3^2-(decomposes to give CO2 gas)}
Data Table 2. Cation Flame Tests.
| Chemical | Cation | Flame Observations |
| KI | Potassium | LIGHT PINK |
| CaCl2 | Calcium | RED |
| LiOH | Lithium | FUSHA PINK |
| NaCl | Sodium | ORANGE |
| Cu(NO3)2 | Copper | TEAL |
| Unknown #105 | TEAL | |
| Unknown #106 | LIGHT PINK | |
| Unknown #107 | RED |
Data Table 3. Complete Chemical Identification.
| Chemical | Cation | Anion Test Observations | Anion | Complete Chemical Name and Formula |
| Unknown #105 | ||||
| Unknown #106 | ||||
| Unknown #107 |
Just need help answering the ***** question under conclusions. here is my worksheet with information
Record your absorbance measurements for the copper (II) sulfate solution in the table below.
| Wavelength (nm) | Absorbance |
| 400 | 0.00 nm |
| 420 | 0.00 nm |
| 440 | 0.00 nm |
| 460 | 0.00 nm |
| 480 | 0.00 nm |
| 500 | 0.00 nm |
| 520 | 0.00 nm |
| 540 | 0.00 nm |
| 560 | 0.02 nm |
| 580 | 0.05 nm |
| 600 | 0.09 nm |
| 620 | 0.14 nm |
| 640 | 0.22 nm |
| 660 | 0.35 nm |
| 680 | 0.50 nm |
| 700 | 0.63 nm |
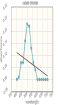
Record your absorbance measurements for the cobalt (II) chloride solution in the table below.
| Wavelength (nm) | Absorbance |
| 400 | 0.00 nm |
| 420 | 0.1 nm |
| 440 | 0.07 nm |
| 460 | 0.07 nm |
| 480 | 0.16 nm |
| 500 | 0.23 nm |
| 520 | 0.22 nm |
| 540 | 0.13 nm |
| 560 | 0.07 nm |
| 580 | 0.04 nm |
| 600 | 0.00 nm |
| 620 | 0.00 nm |
| 640 | 0.00 nm |
| 660 | 0.00 nm |
| 680 | 0.00 nm |
| 700 | 0.00 nm |
Data Analysis
Create and save a graph of absorbance (y-axis) versus the wavelength (x-axis) for both copper (II) sulfate and cobalt (II) chloride. Click the graphing icon below to create your graph. 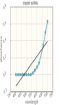
Experiment 2: Measure Absorbance versus Concentration for Cu+2 Ions
Lab Results
What wavelength did you use to measure the absorbance of the copper (II) sulfate solutions?
| 700 nm |
Data Analysis
Create and save a graph of absorbance (y-axis) versus the concentration of the copper (II) sulfate solutions (x-axis). Click the graphing icon below to create your graph. 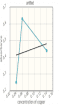
The y-intercept of the plot of absorbance versus concentration is approximately 0. Why is this?
| It should be a straight line obtained by having a slope of = 1 and y-intercept = 0. |
Experiment 3: Determine the Cu+2 Concentration in Unknown Solutions of Copper (II) Sulfate
Lab Results
How many mL of the unknown solutions did you use for your absorbance measurements?
| 10 mL |
Data Analysis
Determine the copper (II) sulfate concentrations in your two unknown samples from the calibration plot.
| wavelength used: 700 nm absorbance unknown: a. 0.08 b. 0.12 |
Conclusions
*******Were the unknown solutions more or less concentrated than the copper (II) sulfate solution used in experiment 1?
Given the data in the table below, what is the concentration of the unknown solution? Both solutions contain the same substance.
| standard solution concentration | 1.25 M |
| standard solution absorbance | 0.75 |
| unknown solution absorbance | 0.38 |
| 0.63 M is the concentration of the unknown solution. |
