CHM135H1 Lecture Notes - Sigmoid Function, Titration Curve, Phenolphthalein

CHM135H1 Full Course Notes
Document Summary
Get access


Related Documents
Related Questions
| Trial 1 | Trial 2 | |
| Volume of Unknown Weak Acid | 4 mL | 4 mL |
| Total Number of Drops Required to Reach Equivalence Point | 5 | 5 |
| Average Number of Drops | 5 | |
| Drops NaOH Added | pH Value Trial 1 | pH Value Trial 2 | pH Value (Average) |
|---|---|---|---|
| Half-Equivalent Point = (46 Drops) | 2.5 | 2.5 | 2.5 |
| Equivalent Point = (93 Drops) | 5 | 5 | 5 |
| 0 | 3.5 | 3.5 | 3.5 |
| 10 | 4.5 | 4.5 | 4.5 |
| 20 | 5.5 | 5.5 | 5.5 |
| 30 | 7.0 | 7.0 | 7.0 |
| 40 | 8.0 | 8.0 | 8.0 |
| 50 | 10.0 | 10.0 | 10.0 |
| 60 | 10.0 | 10.0 | 10.0 |
| 70 | 10.0 | 10.0 | 10.0 |
| 80 | 10.0 | 10.0 | 10.0 |
| 90 | 10.0 | 10.0 | 10.0 |
| 100 | 10.0 | 10.0 | 10.0 |
| 110 | 10.0 | 10.0 | 10.0 |
| 120 | 10.0 | 10.0 | 10.0 |
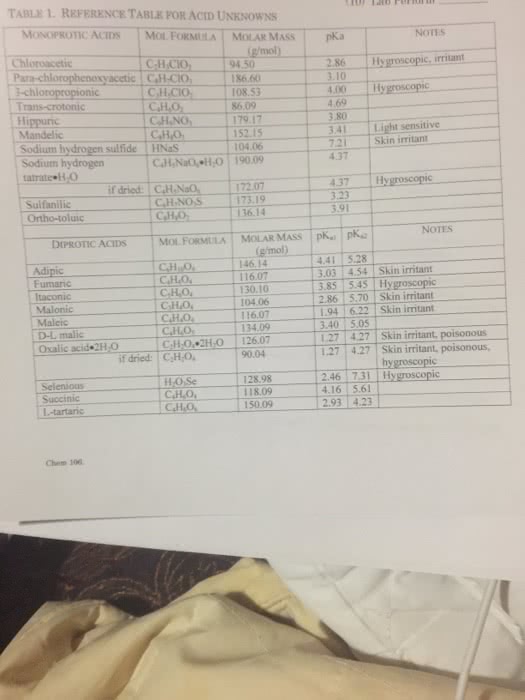
| pKa of Unknown Weak Acid: | 2.5 |
| Ka of Unknown Weak Acid: | 3.16 x 10 ^-3 |
| Unknown Weak Acid Identity: | Citric Acid |
| % Error pKa: | 10.39% |
| % Error Ka: | 4.9% |
A. pH indicator strips work by changing color in the presence of solutions with varied pH values. Thinking about your procedure steps and results in Part 1, why do you think the pH of the unknown weak acid was not determined with pH indicator strips until Part 2.
B. Discuss possible causes of error in the experimental procedure. Why do you think there was a percent of error in the pKa and Ka values of the unknown weak acid, in comparison to the values presented in Table 2?
C. Do you think using a pH meter instead of pH indicator strips would have created a larger or smaller percent error? Explain your answer.
D. Why was phenolphthalein a good indicator to use for determining the equivalence point between the unknown weak acid and strong base?