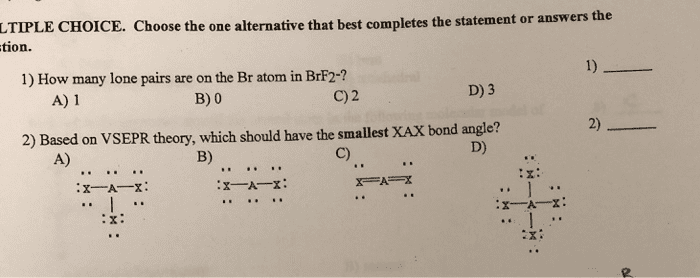CHMA11H3 Study Guide - Midterm Guide: Lone Pair, Sulfur Dioxide, Collision Frequency
125 views6 pages

36
CHMA11H3 Full Course Notes
Verified Note
36 documents
Document Summary
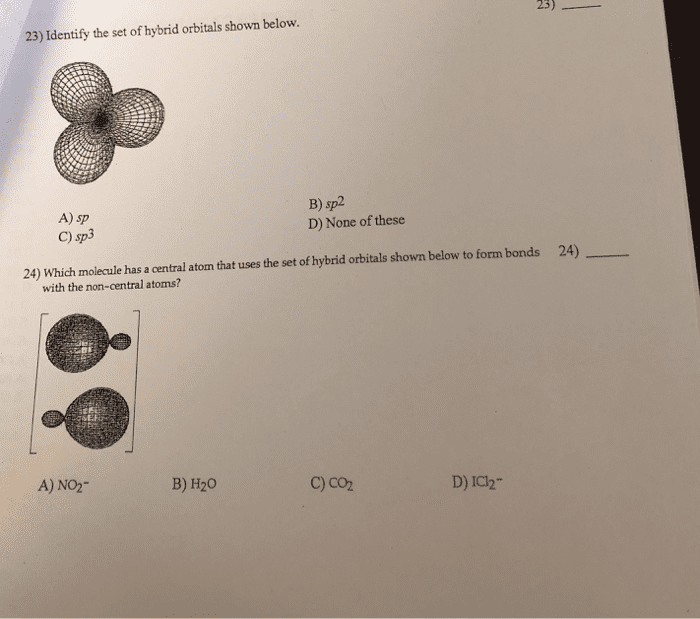
Ch3ch2ch2oh; ii 1,2-propanediol, ch3ch(oh) ch2oh; iii 1,2,3-propanetriol, (glycerol), hoch2ch(oh)ch2oh a) 2so2(g) + o2(g) 2so3(g) ho = -196. 6 kj/mol. The equilibrium is displaced to the left if: the temperature is raised, some sulfur dioxide is added, the pressure is raised, some sulfur trioxide is removed, none of these, write the equilibrium constant expression for the following reaction: 6co2(g) + 6 h2o(l) c6h12o6(s) + 6o2(g) [co2: the relationship between the vapor pressure of a liquid and temperature can be expressed by the clausius-clapeyron equation: Ethanol (c2h5oh) has a normal boiling point of 78. 3oc and, with hvap = 39. 3 kj/mol. [vapor pressure of water at 37oc = 48. 2 torr] a)82: 90% c)18% E)0: henry"s law constants for aqueous solutions at 25oc are 8. 20 x 10-7 molal/mmhg for n2 and. A)2. 04 x 103: none of these, 3. 68 x 104, 8. 05, 6. 13 x 103, for the reaction: 2n2o5(g) 4no29g) + o2(g) the rate law is:
Get access
Grade+20% off
$8 USD/m$10 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
40 Verified Answers