CHEM 121 Lecture Notes - Azimuthal Quantum Number, Atomic Spectroscopy, Photon
13 views2 pages

79
CHEM 121 Full Course Notes
Verified Note
79 documents
Document Summary
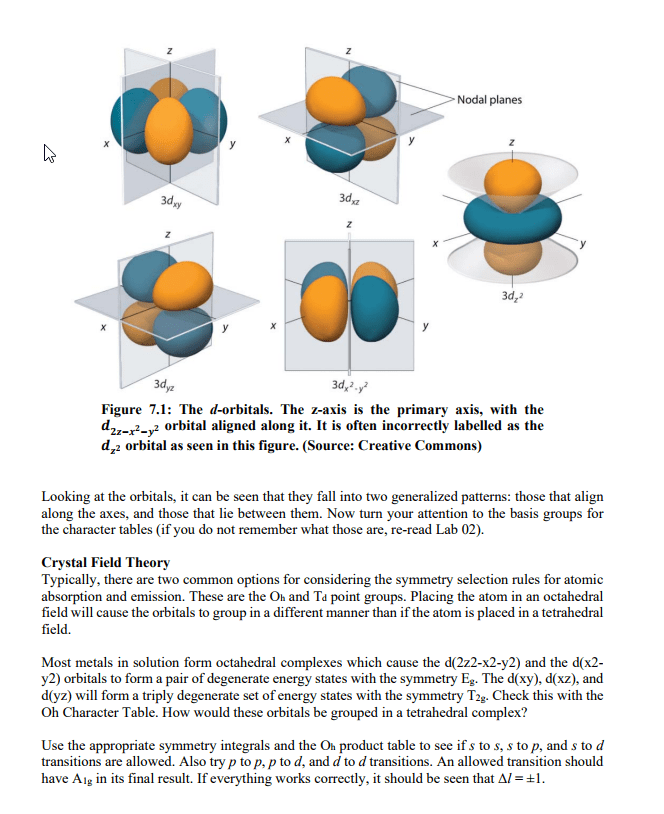
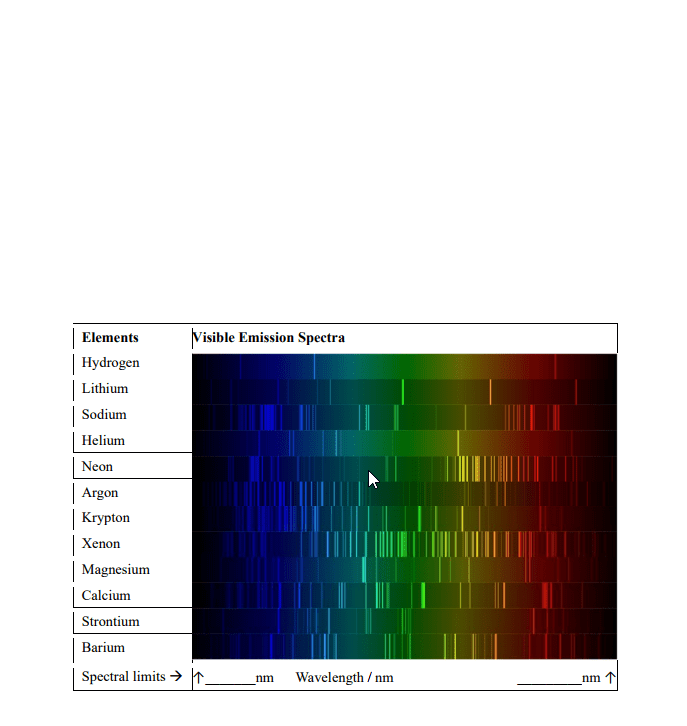
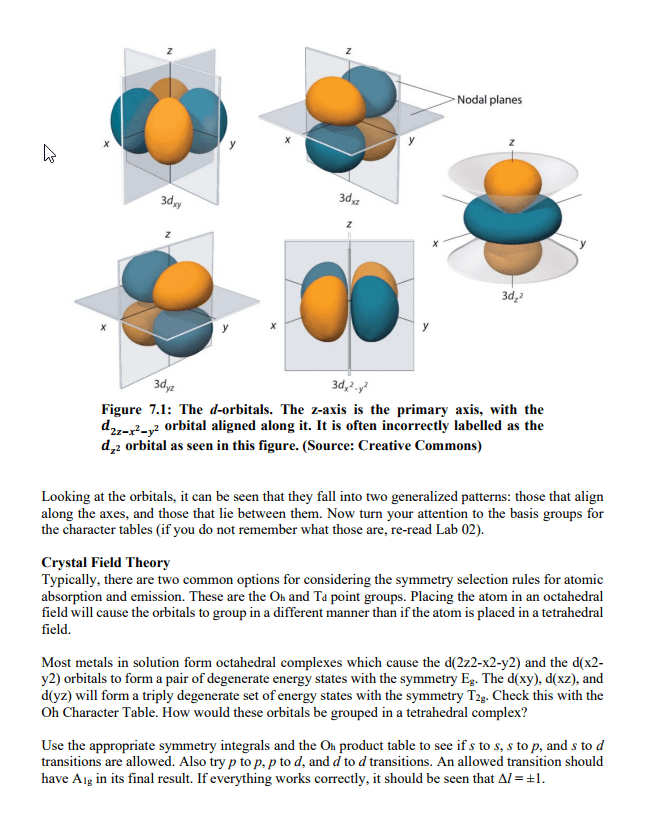
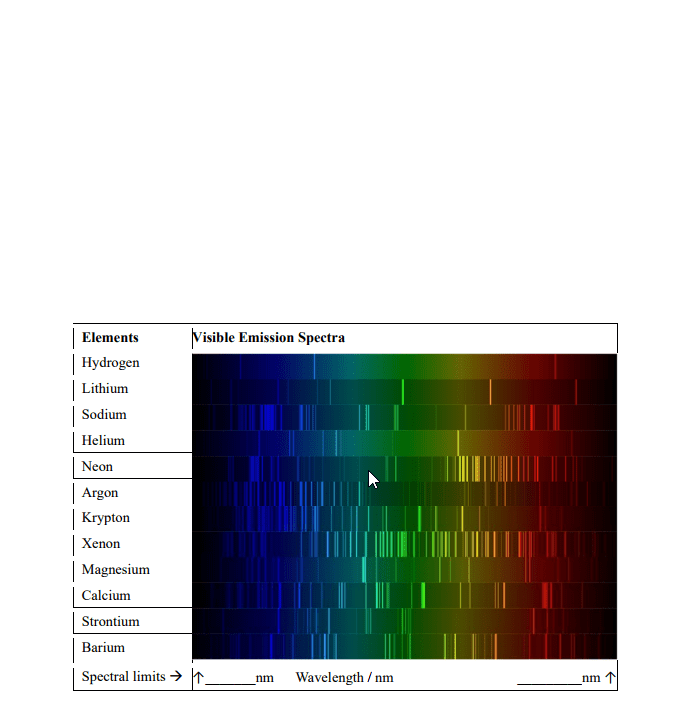
N = nf - ni = anything consistent with energy conservation. When an atom or ion is excited or de-excited in an absorption or emission experiment an electron undergoes a change of state from one orbital to another. The two orbitals involved are described by a set of four quantum numbers, n, l, ml and ms. The first consideration in deciding which transitions are allowed is that of conservation of energy. This requires that the energy of the photon (ephoton = h photon ) is equal to the energy difference ( e) between the two orbitals is the same as the energy of the photon. Therefore, the principal quantum number can change by any amount consistent with the energy change of the transition. This principle leads us to our first selection rule. A selection rule is a statement about the transitions that are allowed.
Get access
Grade+20% off
$8 USD/m$10 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
40 Verified Answers
Class+
$8 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
30 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232