CHEM 343 Lecture Notes - Protecting Group, Aldehyde, Ketone
Document Summary
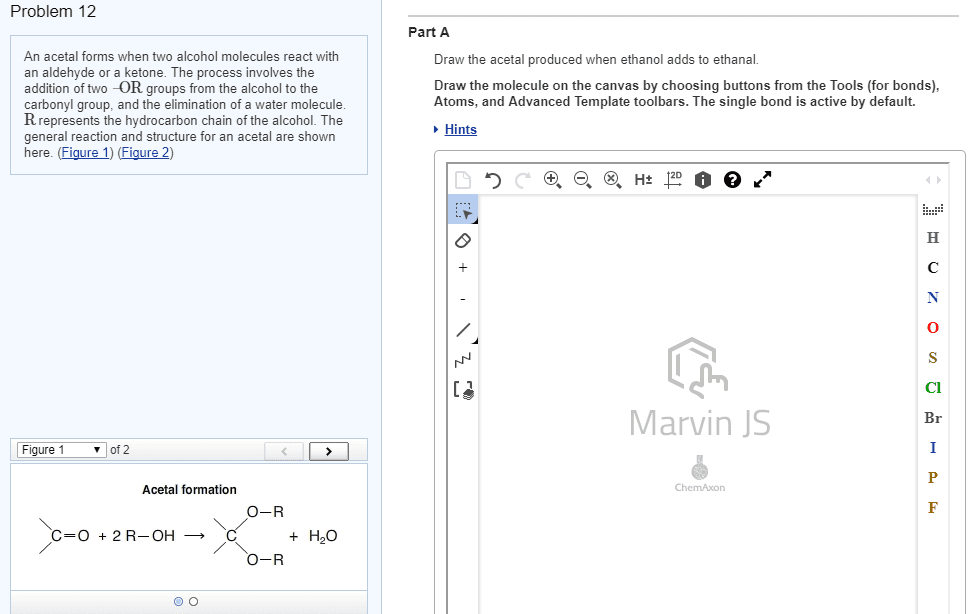
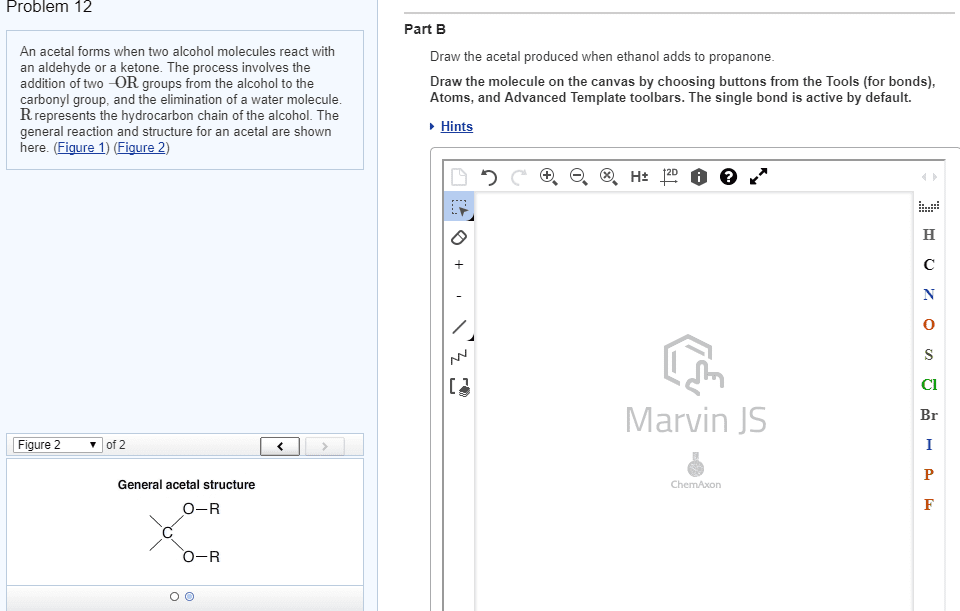
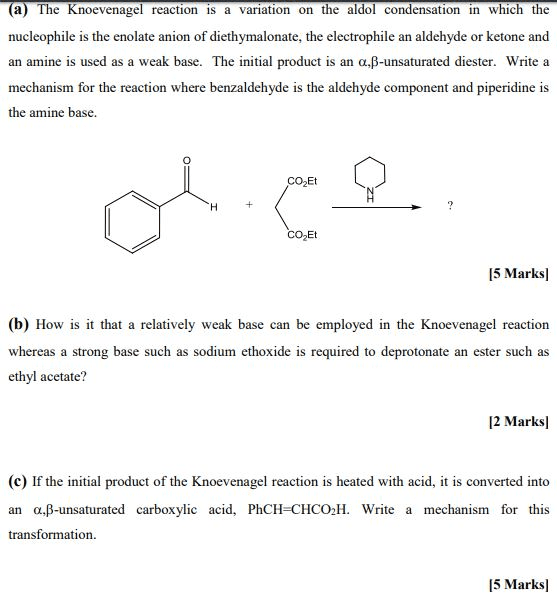
Reductive amination the reaction of an aldehyde or a ketone with ammonia (or with an amine) in the presence of a reducing agent. Hydrate a molecule with two oh groups on the same carbon (gem-diols) The extent to which an aldehyde or a ketone is hydrated in an aqueous solution depends on the aldehyde or ketone. Hemiacetal (hemiketal) the product of the addition of one equivalent of an alcohol to an aldehyde (ketone) Acetal (ketal) the product formed when a second equivalent of an alcohol is added. Acetal (or ketal) formation requires an acid catalyst. Protects a functional group from a synthetic operation that it would otherwise not survive. The c-s bond of thioacetal (or thioketal) can be converted into c-h bonds using h2 and. The wittig reaction a reaction in which an aldehyde or ketone reacts with a phosphonium ylide to form an alkene. Ylide a compound that has opposite charges on adjacent, covalently bonded atoms with complete octets.