CHEM101 Chapter : Pre-Lecture Notes Sept. 13.pdf

77
CHEM101 Full Course Notes
Verified Note
77 documents
Document Summary
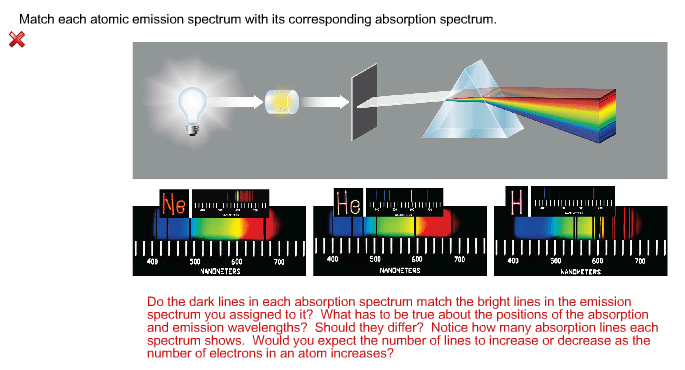
The figure below represents the emission spectrum of a one electron ion in the gas phase. All lines result from transitions to the ground state. Line a has a wavelength of 6. 1 nm. 4 (a) what electronic transition corresponds to line a? (b) identify the ion that exhibits the spectrum. Bohr"s model works very well for h atom and h like atoms. Not so well for atoms with more than 1 e . View the e bound to the nucleus as a standing wave. A string guitar is an example of a one dimension standing wave. Crests and troughs occur at fixed positions. Instead of describing the exact position of the electron, a wave function is used to describe the behavior of the electron. A solution to the schr dinger equation yields . I will not hold you responsible for the equation because the math solution is too complex.