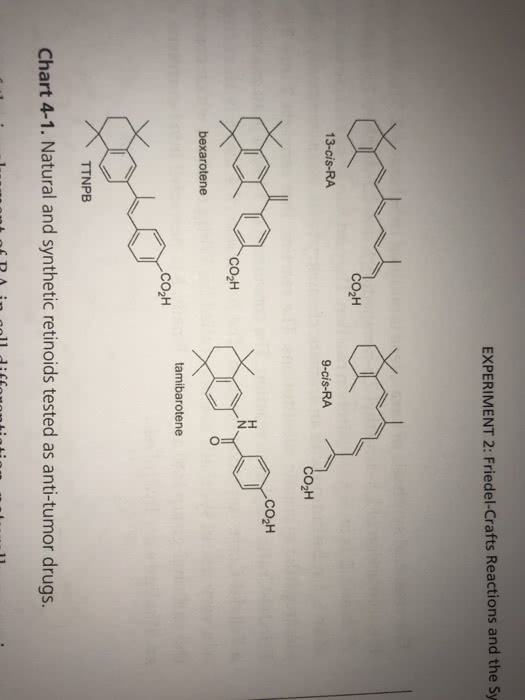
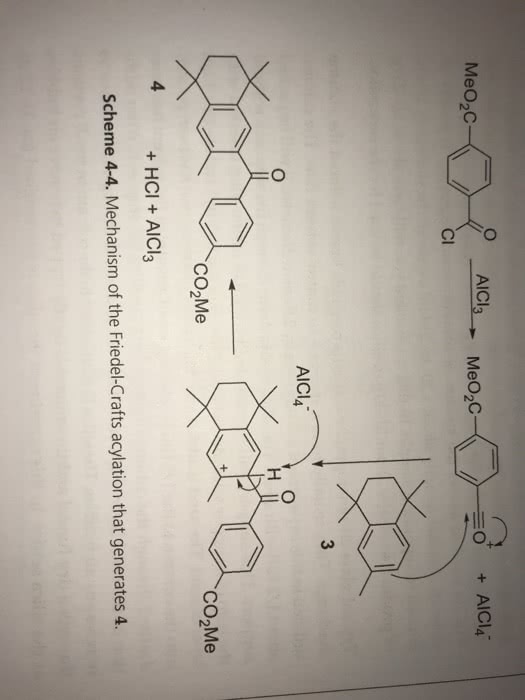
21- Which of these statements is false? A.The Benedict's test allows us to detect the presence of reducing sugars B. The Benedict's test allows us to detect the presence of sugars with a free aldehyde or ketone C. All monosaccharides are reducing sugars because they all have a free reactive carbonyl group. D. All disaccharides have exposed carbonyl groups and are also reducing sugars. E. Sucrose and starches are non-reducing sugars and will not react with Benedict's solution. 22-Select the incorrect statement below about Fehling's test A. Fehling's test is specifically a test for carbohydrates, thus explaining its extensive use itn carbohydrate chemistry B. A positive Fehling's test result is indicated by the formation of a brick red precipitate. C. Fehling's test takes advantage of the ready reactivity of aldehydes by using the weak oxidizing agent cupric ion (Cu2+) in alkaline solution D. In addition to the copper ion, Fehling's solution contains tartrate ion as a complexing agent in the form of copper(II) D-tartrate to keep the copper ion in solution. E. Fehling's reagent is commonly used for reducing sugars but is not specific for aldehydes. F Glucose is a reducing sugar that reacts with Fehling's reagent to form an orange to red precipitate. 23 Select the incorrect statement below about Benedict's test A. Benedict's test can be used to determine the presence of glucose in urine. B. The amount of cuprous oxide formed in Benedict's test is proportional to the amount of reducing sugar present in the urine. C. In Benedict's test, low to moderate levels of reducing sugar turn the solution green; solutions with high glucose levels turn Benedict's yellow or brick red D. Tests using the enzyme glucose oxidase are more specific for glucose than the Benedict's or Fehling's test. E. none of the above 24. Select the incorrect statement below A. Sugars that contain aldehyde groups that are oxidised to carboxylic acids are classified as reducing sugars B. Reducing sugars reduce the Cu2+ in Benedict's solution to Cut which forms as a red precipitate, copper (I) oxide. C. Any sugar that contains a hemi-acetal will be a reducing sugar D. Glycosides are acetals and therefore reducing sugars. E Because the acetals of monosaccharides can undergo mutarotation or oxidation, this means that acetals are nonreducing sugars. F. Fructose (though a ketone) is also a reducing sugar because in the open-chain form, a rearrangement between the hydroxyl group on carbon 1 and the ketone group provides an aldehyde group that can be oxidized. 25. Select the best answer from those below. A. Clinitest is an implementation of the 'Benedict test for reducing sugars. B. Clinitest measures the reducing end concentration of all reducing sugars by comparison with a color charts. C. Clinitest percentage readings are referenced to glucose so that a 1% reducing sugar glucose equivalent corresponds with a 1 gram glucose per 100ml of water concentration. D. All the above are correct