CHEM237L Lecture Notes - Titration Curve, Equivalence Point, Titration
Document Summary
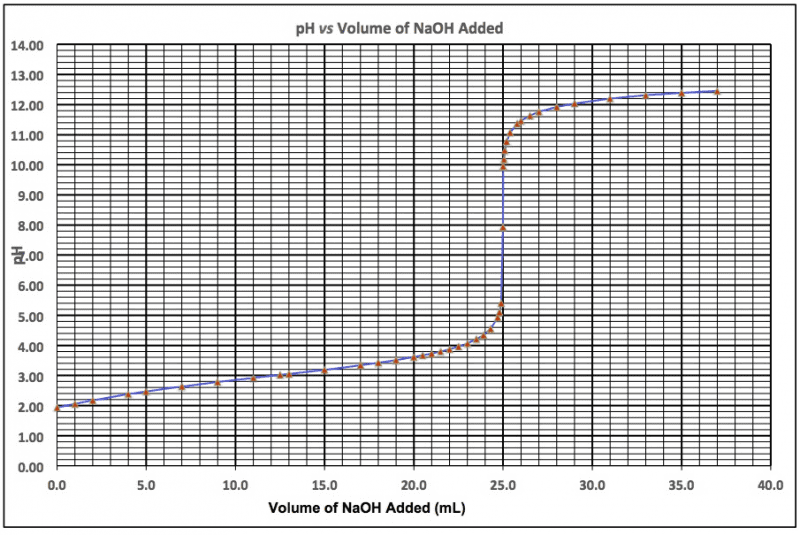
Titration curve of unknown amino acid #34 - trial 1. Buffer point at which excess hcl is neutralized (0. 946 mmol) Titration curve of unknown amino acid #34 - trial 2. Zone point at which excess hcl is neutralized (1. 088 mmol) First derivative plot for trial 1 d e d d a. 4. 726 mmol oh- d e d d a o m m. 8. 0 8. 2 8. 4 8. 6 8. 8 mmol oh added. 2nd equivalence point is at 8. 506 mmol oh- Sample calculations: to get mmol of oh- added, to get ph/ mmol oh added, determining pka values: To determine the ph, the value was estimated by looking at the ph when that amount of mmols is added. An average of the two ph values is taken to result in the experimental ph value. To calculate the first pka value, we know that both pka"s are equal distance from the first. Q-1. (a) the pka values were determined directly from the titration curves.