CHEM 1040 Final: Chemistry exam notes
Document Summary
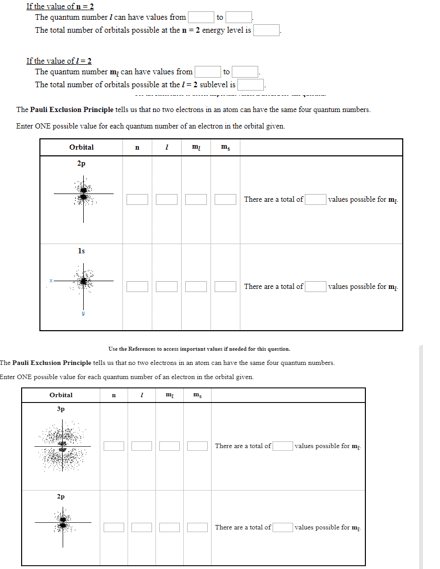
Quantum numbers: principle quantum # (n)- represents main energy level of an atom"s electrons by a number. *increasing value of n corresponds to increasing values of energy for the electron {n i | n>0} n: 1, 2, 3, 4. Angular momentum (l): tells about shape of electron cloud/orbital; l=0-sperical, l=1-dumbell shaped, l=2= 5 variations of shapes. Defines orbital l can equal values from 0--> (n-1) ex. n=2 l=0,1. Spin quantum # ( ): not from shrodinger" s equations, limits # of spin energies for an electron to 2 values 1/2; defines electrons while other q#"s define orbital. Pauli exclusion principle- no 2 electrons can have all 4 q#"s the same; 3 can be the same 1 must be different. Hund"s rule- electrons distributed in sub shell to yield maximum unpaired electrons. Spin pair- an orbital is full- 2 electrons spinning opposite directions ( 1/2)