CHMA11H3 Lecture 7: CHMA11 Lecture 7

36
CHMA11H3 Full Course Notes
Verified Note
36 documents
Document Summary
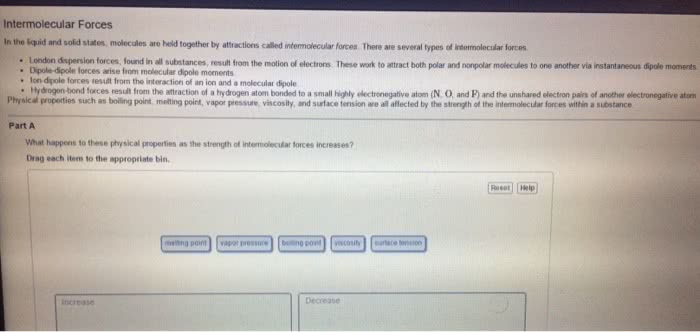
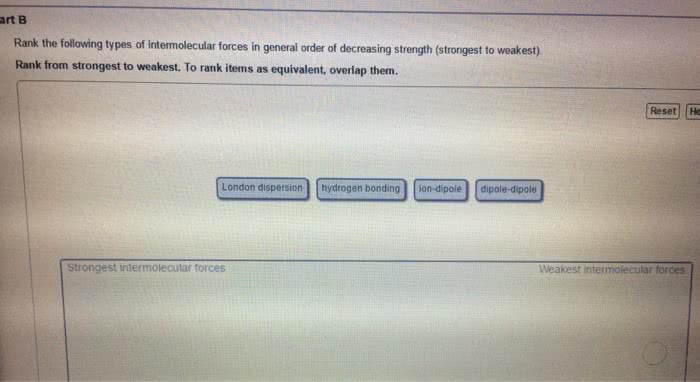
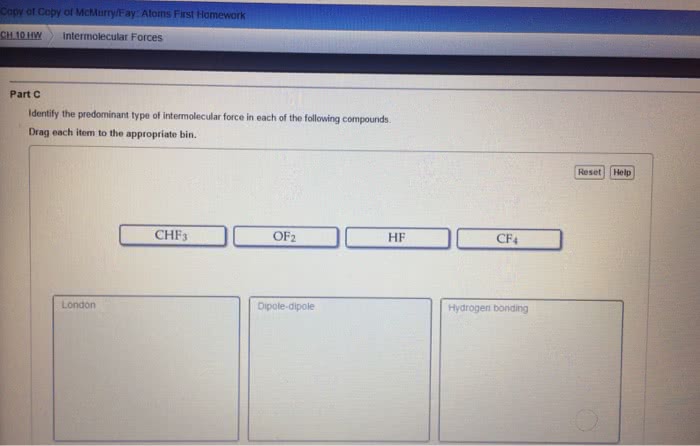
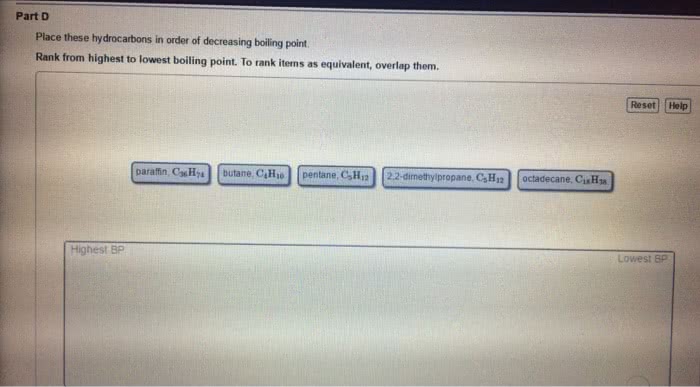
Chma11 lecture 7: chapter 12 crystal structures, solutions and their physical properties. Solutions is a mixture of one thing or several things in one. Greatest component of a solution is the solvent stuff in the solvent is the solute. If we want to make a homogeneous, you have to consider the spontaneity of nature to mix things up and the intermolecular forces that are present. Happens without external forces that causes unmixed things to become more mixed. Conceptual way to look at entropy: state of randomness system that has gained entropy is more random. Ideal gases don"t attract or repel one another they have no intermolecular interactions collide and bounce of each other has no energy reasons to do anything at all. If you compress an ideal gas, it will never make a liquid. Putting two gases together results in them mixing up in totally random ways.