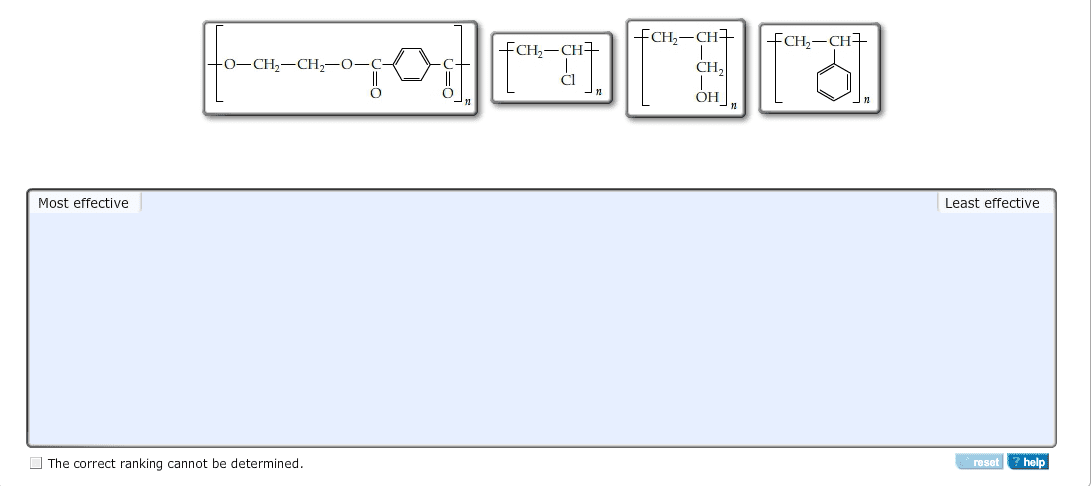
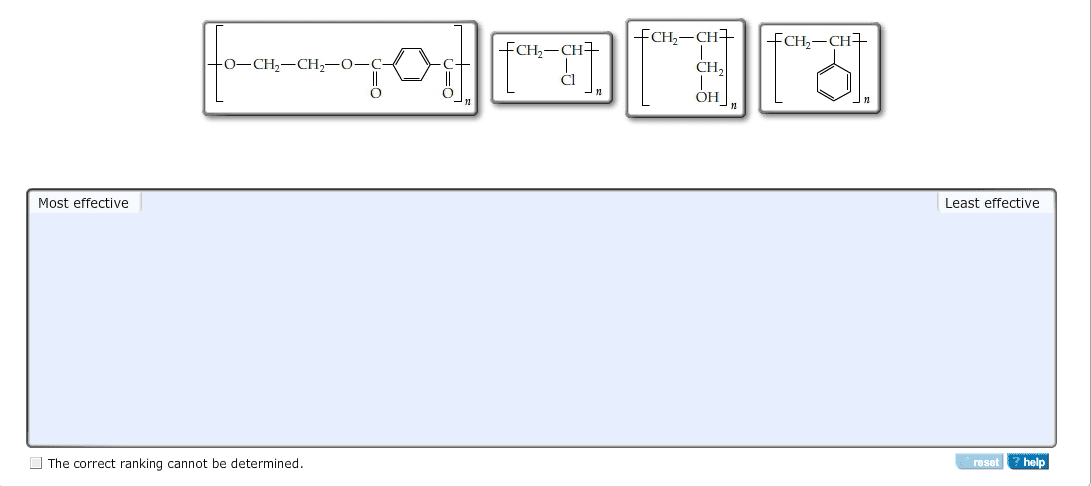
CHEM 110 Lecture Notes - Valence Electron, Metal, Thermal Conductivity

37
CHEM 110 Full Course Notes
Verified Note
37 documents
Document Summary
The challenge to a bonding theory for metals is to explain how bonding can occur with so few electrons. Few valence electrons and many empty orbitals. Metals in crystals surrounded by many neighboring atoms, regular array of the nuclei. For instance, the electron-sea model; is a network of positive ions immersed in a sea of electrons (electrons move freely; they are mobile among array of positive charges) Free electrons are not localized, therefore they are able to absorb photos of visible light, unlike electrons bound to an atom. This is why metals are opaque, they absorb visible light. Electrons at the surface of a metal are able to reradiate (at the same frequency) light that strikes the surface, which explains lustrous appearance of metals. Since electrons are not localized within a bond, they are extremely mobile (they can hop and move) Electricity comes in one side and hops out the other (electron in, electron out)