Experiments performed with biological systems require a buffer that will not interfere with the stability and activities of the biological components. In 1966, Norman Good and his colleagues began a series of investigations that established criteria for new zwitterionic buffers that would be optimal for experimentation with biological systems.
These criteria include:
a. pKa between 6 and 8
b. highly water soluble
c. minimally permeable to biological membranes
d. minimal effects of salt concentration
e. minimla effect of temperature on pKa
f. minimal effect of concentration on pKa
g. minimal interaction with metal ions
h. nontoxic
i. high chemical stability
j. minimal absorbance between 240-700 nm
For each buffer below, choose the property tha does NOT meet Good's criteria for biochemical buffers. If all of the buffer's properties satisfy the criteria for a Good's buffer, then select the last choice. 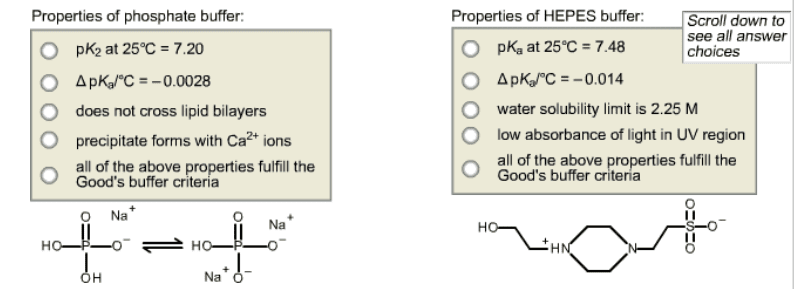
Properties of phosphate buffer: O pK2 at 25°C 7.200 O ApKaleC 0.0028 O does not cross lipid bilayers O precipitate forms with Ca Ons o all of the above properties fulfill the Good's buffer criteria O Na Na Na Properties of HEPES buffer Scroll down to see all answer O pKa at 25°C 7.48 choices O ApKoloC 0.014 O water solubility limit is 2.25 M O low absorbance of light in UV region o all of the above properties fulfill the Good's buffer criteria



