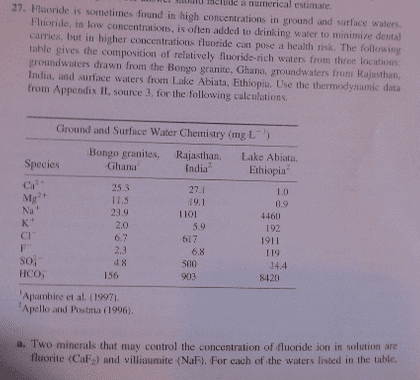

Fluoride is sometimes found in high concentrations in ground and surface waters. Fluoride, in low concentrations, is often added to drinking water to minimize dental carries, but in higher concentrations fluoride can pose a health risk. The following table gives the compositions of relatively fluoride-rich waters from three locations ground waters drawn from the Bongo granite, Ghana, ground waters from Rajasthan, India. and surface waters from Lake Abiata, Ethiopia. Use the thermodynamic data from Appendix II, source 3, for the following calculations. 1 Apambire et al. (1997) 2 Apello and Postana (1996). Two minerals that may control the concentration of fluoride ion in solution are fluorite (CaF2) and villiaumite (NaF). For each of the waters listed in the table. determine the saturation index at 25 degree C for these minerals. (Note: Activity is important and must be included in the calculations,) Would either of these minerals control the fluoride concentration of these waters? What is a possible explanation for the very high concentrations of Na + , CI - , F - and HCO in the waters of Lake Abiata? The lake has a pH of 9.62.