CHMA10H3 Chapter 1: Chapter 1 – Matter, Measurement and Problem Solving

36
CHMA10H3 Full Course Notes
Verified Note
36 documents
Document Summary
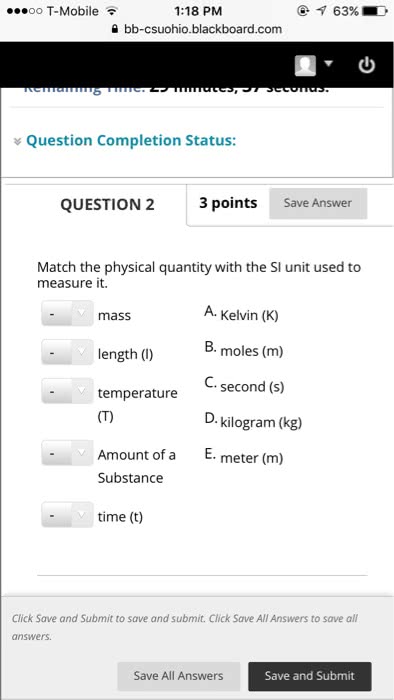
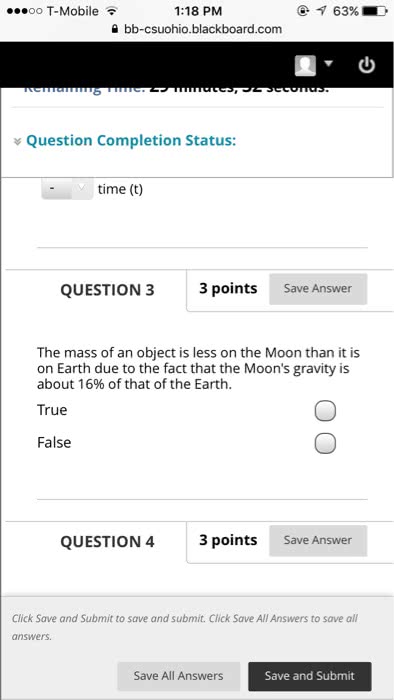
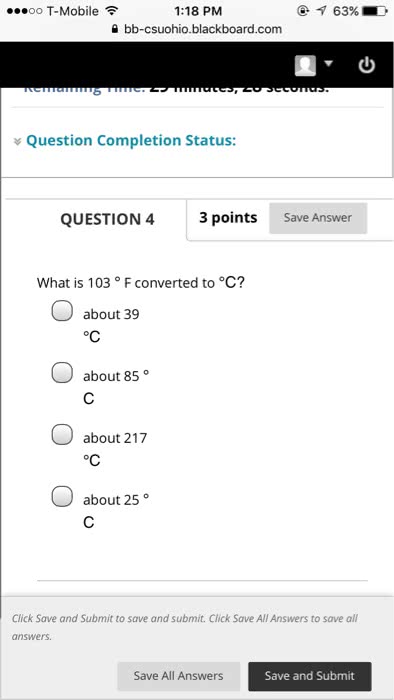
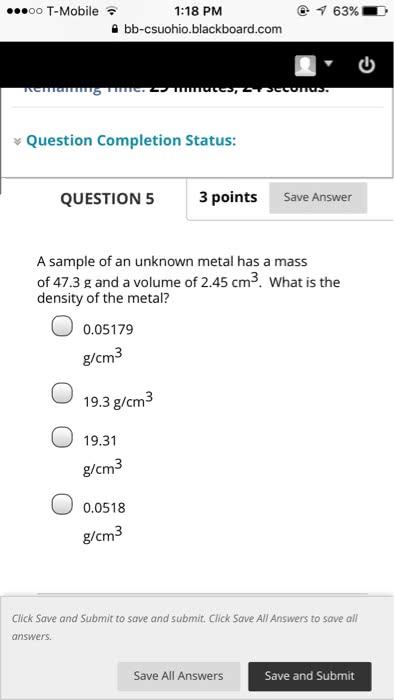
Chapter 1 matter, measurement and problem solving. The properties of matter are determined by properties of atoms (submicroscopic particles that makes fundamental building block for matter) and molecules (2 or more atoms joined in specific geometrical arrangement) Chemistry: science that seeks to understand behavior of matter by studying behavior of atoms and molecules. Scientific knowledge is empirical (based on observation and experiment) leads to hypothesis (tentative explanation of observation) test hypothesis through experiments (highly controlled procedures to generate observations and see if hypothesis is wrong or correct) Series of similar observations can lead to scientific law (brief statement that summarizes past observation and predicts future ones) or a theory (model for the way nature is and t ries to explain what and why) In summary, this is called the scientific method. Example: law of conservation of mass (antoine lavoisier) in a chemical reaction, matter is neither created nor destroyed (a law summarizes a series of related.