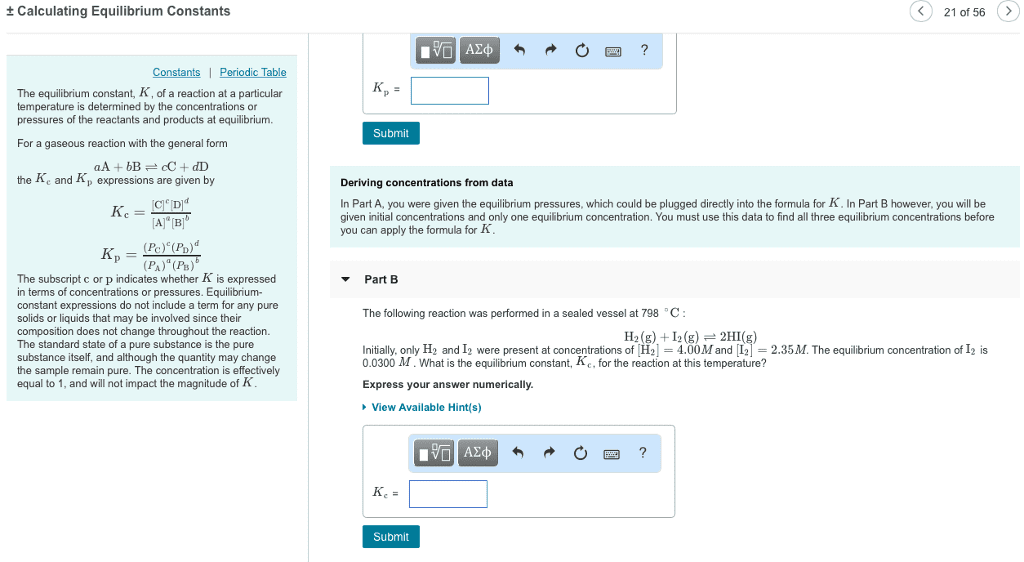
CHEM1101 Study Guide - Midterm Guide: Sodium Hydroxide, Activation Energy, Ammonia

Homogeneous vs. Heterogeneous
• Hoogeeous euiliiaou he all eatats ad poduts ae i the sae phase.
• Heteogeeous euiliiaou he soethig i the euiliiu is i a diffeet phase. • The
value used for the concentration of a pure substance is always 1.
The Decomposition of CaCO3— A Heterogeneous Equilibrium
• The euatio fo the eatio is CaCO3s ⇌CaO(s) + CO2(g)
• This results in Kc = [CO2] and Kp = PCO2
Two comments about Kc
• The oetatio of pue liuids eg liuid ate ad solids do ot appea o i the K
expression.
• The ueial alue of K is eaigful ol he it is assoiated ith a patiula hemical
equation.
Is a Mixture in Equilibrium? Which Way Does the Reaction Go?
• To ase these uestios, e alulate the eatio uotiet, Q.
• Q looks like the euiliiu ostat, K, ut the alues used to alulate it ae the uet
conditions, not necessarily those for equilibrium.
• To alulate Q, oe sustitutes the iitial oetatios of eatats ad poduts ito the
equilibrium expression.
Comparing Q and K
• Natue ats Q = K (equilibrium)
• If Q < K, atue ill ake the eatio proceed to products.
• If Q = K, the eatio is i euiliiu.
• If Q > K, atue ill ake the eatio poeed to eatats.
Le Châtelier’s Principle
If a sste at euiliiu is distued a hage i tepeatue, pessue, o the oetatio
of one of the components, the system will shift its equilibrium position so as to counteract the effect
of the distuae.
Change in Reactant or Product Concentration
• If the sste is i euiliiu –adding a reaction
component will result in some of it being used up. –
removing a reaction component will result in some if
it being produced.
find more resources at oneclass.com
find more resources at oneclass.com