CHEM1100 Study Guide - Boiling Point, Gibbs Free Energy, Lattice Energy
Document Summary
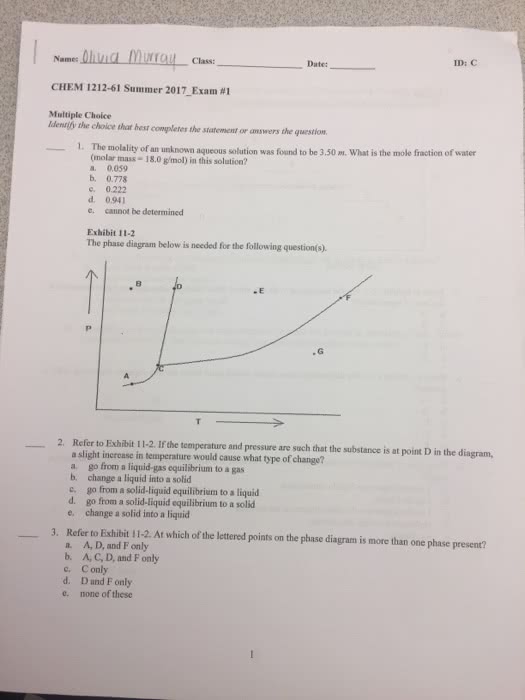
Heterogenous equilibria: reactions where reactants and products exist as more than one phase. Equilibrium constant does not include pure solids or liquids as these are constants. Pressure is not affected by adding more liquids or solids. Phase change: transition of substance from one phase to another (no chemical reaction) Depend on temperature, pressure and magnitudes of intermolecular forces. Phase changes require that energy is supplied or removed from the substance undergoing the change. Molar enthalpy of fusion: heat required to melt 1 mol of substance at its normal melting point. Molar enthalpy of vaporisation: heat required to vaporise 1 mol of substance at its normal boiling point. I. e. sweat (an endothermic process) has a cooling effect. Molar enthalpy of sublimation: heat required to vaporise 1 mol of substance from solid phase. Triple point: where all three phases are in equilibrium. Supercritical fluid: form upon compression of gases at high temperature or heating a liquid to high pressure.