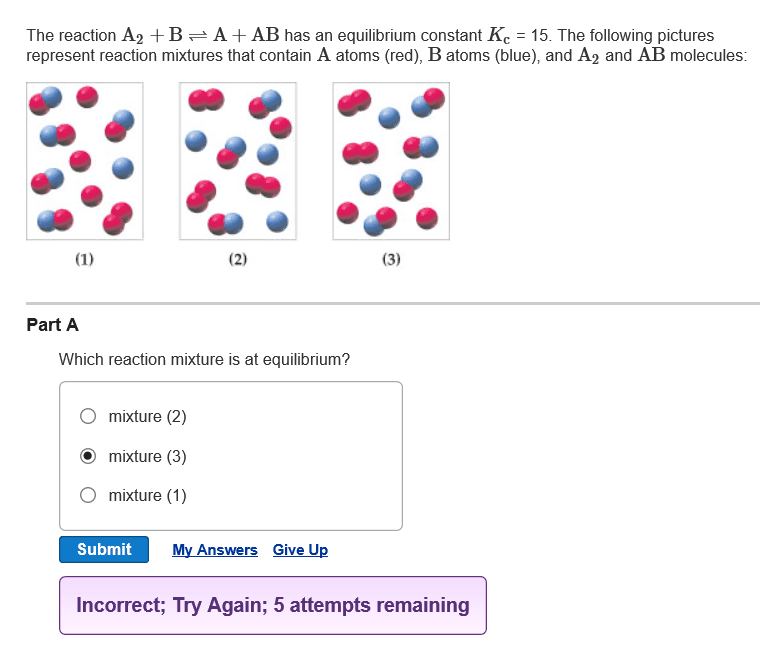
Explain each question plz!
26. If Qc is greater than Kc, then. (A) products will be converted to reactants. (B) reactants will be converted to products (C) the system is at equilibrium. (D) this is an impossible situation 27. The reaction A2 + B2ä¸2 AB has an equilibrium constant KC-64 The pictures below represent reaction mixtures that contain A2 molecules (shaded) and B2 molecules (unshaded), and AB molecules. Which reaction mixture is at equilibrium? 28. At 200°C, the equilibrium constant (Kp) for the reaction below is 2.40 x 10 2NO (8)N (B) O (e) A closed vessel is charged with 36.1 atm of NO. At equilibrium, the partial pressure of O is atm. A. 294 8. 35.7 D. 6.00 E. 1.50 x 10-2 29, The gauilibriumexpression for 4 HCI(g) +02(g)ä¸HOU) +2Cb(g) is (B) Kc=[AHCI]+[02] [C12]2 [H2ì [HCl [O21 HCIn to2l