CHY 103 Study Guide - Midterm Guide: Fluorine, Oxidation State, Potassium Nitrate
Document Summary
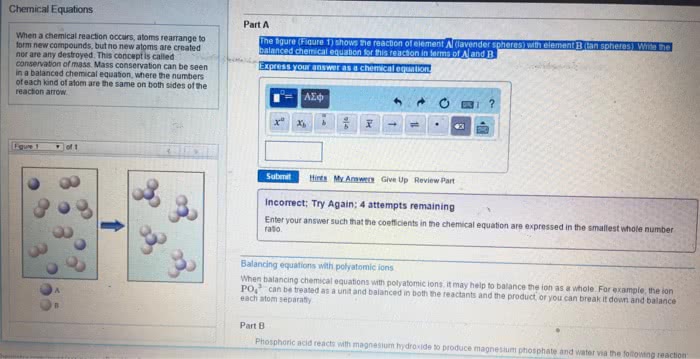
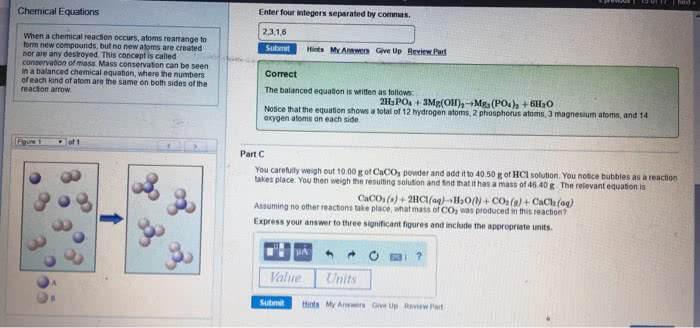
General chemistry chapter 4- chemical reactions and stoichiometry. Co2 is a gas, and it releases which makes the cake puffs. A chemical reaction can be represented by an unbalanced chemical equation, but we need to balance it. Reactants and products must be equal on both side. Always balance atoms that appear on both sides first. 3n2h4 ---> 4nh3 + 3n2 hydrogen is balanced. Oil is an example of non-polar and water is polar. Sugar is slightly polar (not really) (cid:271)e(cid:272)ause there are so(cid:373)e o(cid:454)(cid:455)ge(cid:374) i(cid:374) it, a(cid:374)d it"s a(cid:271)le to dissol(cid:448)e i(cid:374) (cid:449)ater. Nonelectrolyte do not produce any ions, but they still dissolve in solutions. Soluble means being 100% ionzied (aquous means fully soluble) If it"s solu(cid:271)le, it has produ(cid:272)ed io(cid:374)s. e(cid:448)e(cid:374) (cid:449)eak ele(cid:272)trol(cid:455)tes (cid:272)a(cid:374) produ(cid:272)e the(cid:373) (cid:271)ut (cid:449)e (cid:272)all the(cid:373) (cid:449)eak. Salt is a (cid:373)ole(cid:272)ule that is (cid:373)ade of a (cid:373)etal a(cid:374)d (cid:374)o(cid:374)(cid:373)etal, like nacl, the(cid:455)"re io(cid:374)i(cid:272).