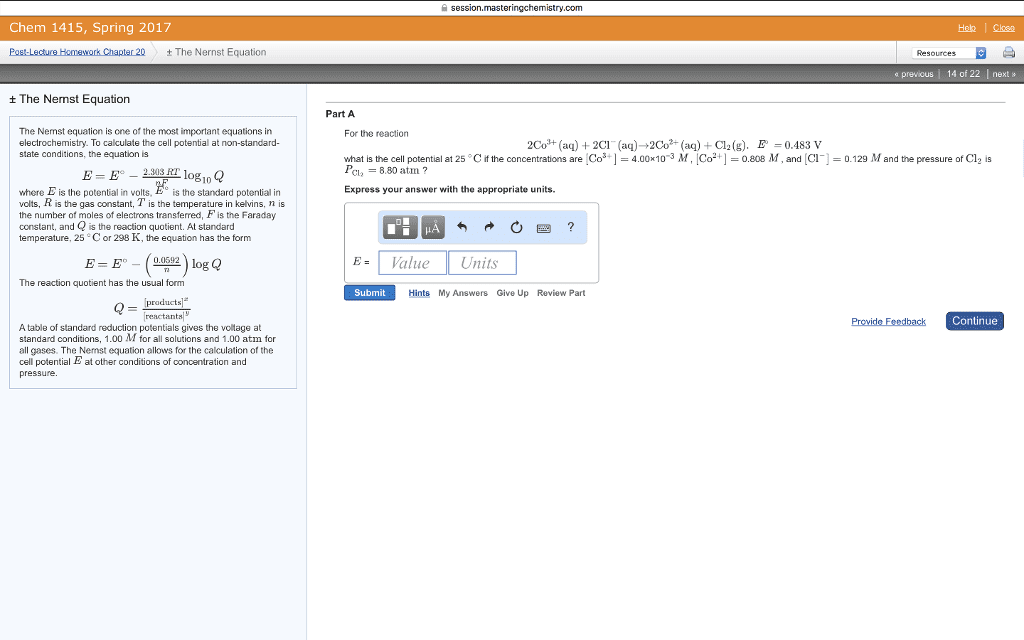
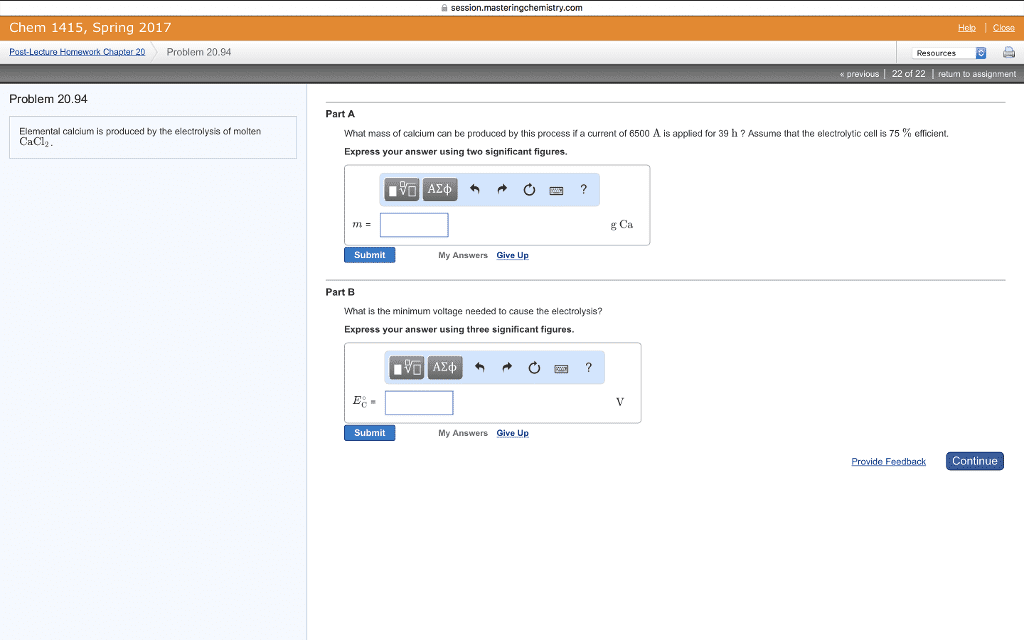
CHEM 1300 Study Guide - Final Guide: Thermodynamic Equilibrium, Equilibrium Constant, Partial Pressure
Get access

Related textbook solutions
Chemistry: Structure and Properties
Basic Chemistry
Principles of Chemistry Molecular Approach
Principles of Chemistry Molecular Approach
Chemistry: Structure and Properties
Chemistry: A Molecular Approach
Chemistry: A Molecular Approach
Principles of Chemistry: A Molecular Approach
Chemistry: The Central Science
Related Documents
Related Questions
answer all questions for rating.
1.
Part A
Phosgene (carbonyl chloride), COCl2, is an extremely toxic gas that is used in manufacturing certain dyes and plastics. Phosgene can be produced by reacting carbon monoxide and chlorine gas at high temperatures:
CO(g)+Cl2(g)âCOCl2(g)
Carbon monoxide and chlorine gas are allowed to react in a sealed vessel at 453 âC . At equilibrium, the concentrations were measured and the following results obtained:
| Gas | Partial Pressure (atm) |
| CO | 0.800 |
| Cl2 | 1.24 |
| COCl2 | 0.240 |
What is the equilibrium constant, Kp, of this reaction?
Part B
The following reaction was performed in a sealed vessel at 737 âC :
H2(g)+I2(g)â2HI(g)
Initially, only H2 and I2 were present at concentrations of [H2]=3.10M and [I2]=2.35M. The equilibrium concentration of I2 is 0.0100 M . What is the equilibrium constant, Kc, for the reaction at this temperature?
2.
Part B
The following reaction was carried out in a 3.25 L reaction vessel at 1100 K:
C(s)+H2O(g)âCO(g)+H2(g)
If during the course of the reaction, the vessel is found to contain 8.00 mol of C, 14.4 mol of H2O, 3.90 mol of CO, and 6.10 mol of H2, what is the reaction quotient Q?
Part C
The reaction
2CH4(g)âC2H2(g)+3H2(g)
has an equilibrium constant of K = 0.154.
If 6.60 mol of CH4, 4.10 mol of C2H2, and 11.50 mol of H2 are added to a reaction vessel with a volume of 5.80 L , what net reaction will occur?
The reaction
has an equilibrium constant of = 0.154.
If 6.60 of , 4.10 of , and 11.50 of are added to a reaction vessel with a volume of 5.80 , what net reaction will occur?
| The reaction will proceed to the left to establish equilibrium. | |
| The reaction will proceed to the right to establish equilibrium. | |
| No further reaction will occur because the reaction is at equilibrium. 3. At 700 K, Kp = 0.140 for the reaction ClF3(g)âClF(g)+F2(g). Calculate the equilibrium partial pressures of ClF3, ClF, and F2 if only ClF3 is present initially, at a partial pressure of 1.59 atm . |