CHEM120L Final: CHEM 120L Experiment 5 Exam Review
847 views2 pages
9 Dec 2016
School
Department
Course
Professor
Document Summary
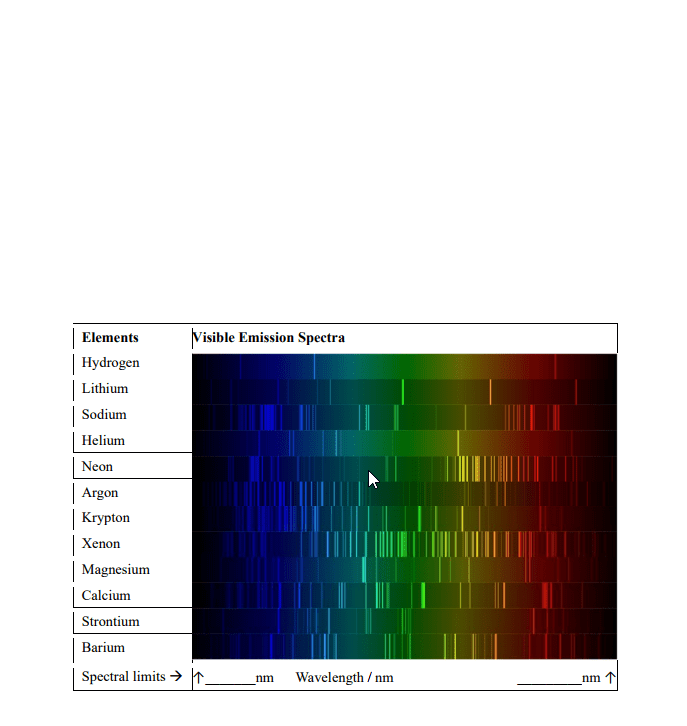
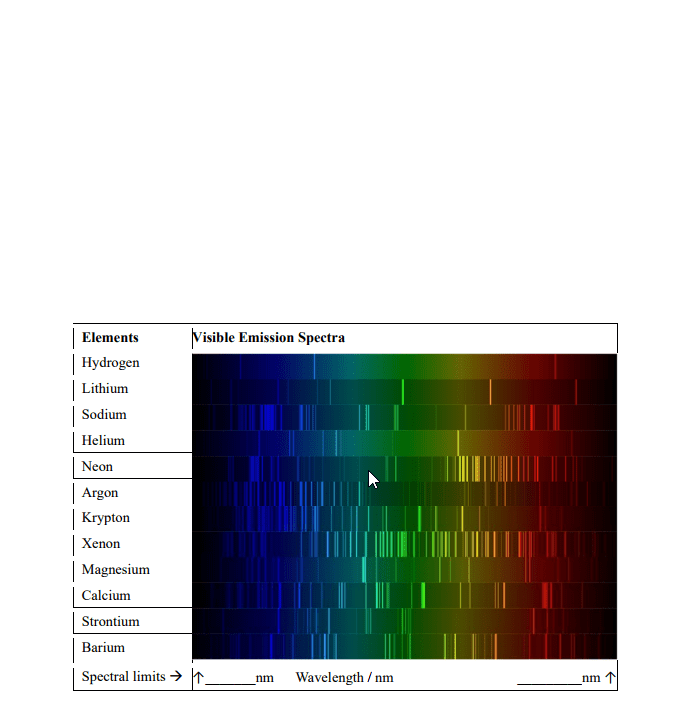
Emission spectra and electronic structure of atoms: the h spectrum. Measure the wavelengths of the visible lines in the hydrogen spectrum using a spectroscope. Visualize emission spectra of salt solutions, k+, ca2+ and li+ When electrons get excited they move to a high energy state (absorb energy). This energy can be provided by heat, light or an electrical discharge. Photons of light are emitted when the atom goes from a higher energy state (nh) to a lower energy state (nl) Balmer series when nl = 2, lies in visible region. Lyman series hydrogen spectral series of transitions, results in uv emission lines. Colour of light emitted by a specific atom depends on how much energy the electron releases as it moves down to a lower energy level. Spectrum set of individual colours emitted by an element. General equation suggested by rydberg in 1890: (cid:1874)= (cid:2869)= (cid:4666) (cid:2869)(cid:3041) .
Get access
Grade+20% off
$8 USD/m$10 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
40 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232